How DMSO Heals the Brain and Transforms Neurology
The extensive evidence behind DMSO's ability to treat 'incurable' neurological diseases — and how to use it
Story at a Glance:
DMSO is an “umbrella remedy” capable of treating a wide range of challenging ailments due to its combination of therapeutic properties (e.g., improving circulation, reducing inflammation, protecting cells from a myriad of otherwise lethal stressors, and reviving dying cells).
These properties make DMSO uniquely suited to treat “incurable” neurological disorders, and in conjunction with forgotten research on the effects of microcirculatory impairments, reshape our understanding of the causes of neurological (and psychiatric) diseases.
DMSO has shown remarkable promise for cognitive impairment, brain fog, and memory loss from a wide range of causes (e.g., aging, vascular disease, anesthesia, post-COVID or pharmaceutical poisoning), along with improving sleep quality and dream vividness — often by resolving the pain, breathing difficulties, or neurological conditions (e.g., restless leg syndrome) that were preventing restorative sleep.
Extensive data supports DMSO’s use for the major neurodegenerative diseases — including Parkinson’s, Alzheimer’s, ALS, Huntington’s, and multiple sclerosis — and protein misfolding diseases such as prion disorders and Niemann-Pick disease, where DMSO’s ability to act as a chemical chaperone (stabilizing proteins and dissolving toxic aggregates) and augment cranial drainage is particularly relevant.
Numerous psychiatric conditions (e.g., schizophrenia, depression, anxiety, and PTSD) have responded to DMSO, as have seizures and epilepsy, movement disorders, encephalitis, myasthenia gravis, and hydrocephalus.
DMSO has also produced striking developmental improvements in children with Down syndrome across multiple clinical studies, along with many other neurodevelopmental disorders.
This article will synopsize the extensive data demonstrating DMSO’s efficacy for CNS neurological diseases (approximately 2000 studies and 200 pertinent reader testimonials), share pertinent (non-DMSO) discoveries we’ve made about neurological diseases over the years, and then conclude with practical guidance on DMSO protocols and complementary approaches that also aid in the treatment of common neurological disorders.
DMSO has remarkable therapeutic utility across a wide range of challenging conditions yet is largely unknown. Recognizing this, I spent the last two years compiling the data which shows DMSO treats a wide array of conditions including lung issues (e.g. COPD and Asthma), skin issues (including hair loss), many different types of pain, arthritis, tissue injury (e.g., sprains or burns), eye issues (e.g., vision loss or dry eyes), autoimmune disorders, dental issues, gastrointestinal diseases, infections, and cancers (along with how DMSO’s efficacy can be further enhanced by combining it with pharmaceuticals or natural therapies).
As each of the above articles, drawing upon thousands of forgotten research studies, made a convincing case to try DMSO, they collectively received millions of views, and thousands of readers (currently over 6000) reported to me that improvements happened across a vast swathe of conditions they were dealing with, many of which were life-changing or match those in this 1980 news program:
More importantly, much in the same way Mike Wallace successfully revived interest in DMSO in 1980 after the FDA successfully spent two decades largely burying it, this series has again created a renewed interest in one of the most accessible and effective remedies available to us.
Since DMSO is particularly well-suited to treating neurological diseases (which often “nothing can be done about”), some of the most profound stories I’ve received related to neurological diseases. Likewise, consider this conversation I had a few days ago with James Miller MD, a physician, who inspired by the results reported here, began using it in a large number of his patients, and frequently seeing astonishing results he was initially left in disbelief by.
JM: Hope you are doing well. Just checking in, haven't seen a lot of your postings lately.
Me: Sorry, I turned into a ghost. I have been working as hard as I can on getting the DMSO neurology article done; there’s just so much to unpack.
JM: It is my impression, with no hard data, that ~80% of everything people see neurologists for goes away with DMSO. That is what my patients reflect back to me who choose to trial DMSO for their neurological problems.
Me: That’s basically why I’ve been working so hard on this.
Likewise, I recently received a remarkable testimonial from an ALS patient (the horrible terminal disease we’ve seen decades of research fundraisers for) that shows there may be real hope for this incurable disease.
Note: if any of you have profound DMSO stories you would like to share, please share them here and consider reaching out to Rebecca so she can document them.
Lastly, for those of you seeking DMSO protocols and recommendations, they can be found at the end of this article (which I advise reading first to better understand those instructions).
Cellular Protection
DMSO is well-known for protecting cells from many otherwise deadly stressors. For example, it prevents freezing damage to cells, which made modern cryopreservation (cryomedicine) possible, and extensive research shows that this protective effect works across nearly every type of tissue (along with repeatedly saving human fingers and animal ears or limbs1,2,3 from being lost to frostbite). As the cells of the nervous system are particularly sensitive to injury (and often unable to heal from it), DMSO’s protective qualities are particularly useful for it. Core mechanisms of protection include:
•Reducing oxidative stress1,2,3,4 and neutralizing harmful free radicals1,2,3 (e.g., those caused by radiation like hydroxyl) through scavenging charged ions (e.g., H+) and forming protective DMSO radicals (along with decreasing lipofuscin formation in human glial cells, reducing the cumulative oxidative damage that drives cellular aging). In hippocampal slices DMSO also counteracted this oxidative stress,1,2 and in cerebellar granule neurons, this prevented oxidative stress-induced apoptosis and cell death by reducing early mitochondrial impairment and DNA fragmentation1,2 (with similar benefits also being seen when DMSO was combined with CDK and G9a inhibitors1,2). Trace amounts of DMSO also protect plants from ozone gas injury and counteract reactive hypochlorous acid, superoxide, and hydrogen peroxide (while simultaneously working synergistically with oxidative therapies and not impairing neutrophil viability).
•Increasing the production of ATP in cells, and facilitating producing it when energy production has been compromised (e.g., minute concentrations of DMSO, as low as 0.000025–0.25%, have been shown to increase cellular metabolism such as by shunting metabolites from glycolysis to the mitochondrial Krebs cycle or to make a part of the mitochondria able to synthesize ATP without the rest of the mitochondria being present1,2,3,4,5). DMSO also prevented hydroxyl radical-induced mitochondrial aconitase inactivation, ATP depletion, and neuronal damage. Furthermore, DMSO increased the metabolism of pyruvate and glucose in brain slices, protected mice from otherwise lethal nitrogen asphyxiation, and in a study where mice were decapitated, DMSO prolonged how long the mice continued to gasp (breathe) and hence how long brain function continued.
Note: many animal studies are exceedingly cruel and not something I support; however, as they have been done, I felt it was important to share the knowledge certain DMSO ones provided so it would not be necessary to repeat them to acquire that data.
•Protecting cells from dying once the blood supply is cut off (a key reason why so many readers have been able to avoid permanent disability from a stroke), including by preventing the rapid influx of calcium or sodium ions that frequently triggers apoptosis (cell death), and reducing the activity of caspase proteins (which trigger cell death) in the liver, heart, and airway epithelial cells.
Note: ⬖ designates natural substances used in conjunction with DMSO.
These properties hence allow DMSO to:
•Protect neurons throughout the brain (e.g., in the hippocampus) from a wide range of excitotoxins—which are well-recognized as a common cause of neurodegeneration,1,2,3,4,5,6,7,8,9,10 (e.g., in one study DMSO restored 66.7-76.1% of normal electrical activity following glutamate toxicity), and to enhance the protective effects of other protective agents (e.g., syringaresinol,⬖ isoquinolinesulfonamides, curcumin⬖ and ginkgo biloba⬖).1,2,3,4,5,6
Note: DMSO is routinely combined with other neuroprotective agents such as curcumin⬖, melatonin,⬖ baicalin,⬖ butein,⬖ icariin,⬖ naringin,⬖ 4-PBA, and BPV(phen) , various Chinese medicinals, nitrone compounds, and capsaicin derivatives⬖ (i.e., in the studies just listed, these combinations reduced neuroinflammation, oxidative stress, ER stress, and apoptosis while enhancing mitochondrial function and autophagy in neuronal cells).
•In carbon monoxide poisoned rats, reducing cerebral neuronal alteration and degenerative rate, along with total cardiac injury score (and also reducing liver injury if combined with ethyl pyruvate).1,2 Glibenclamide further improved neurological deficit scores, reduced neural cell breakdown (NSE and S-100β) and reduced inflammatory TNF-α and IL-8 levels. Lastly, DMSO’s antioxidant properties have been proposed to confer a potential neuroprotective role in carbon monoxide poisoning.1,2
•Protect normal cells against chemotherapies such as preventing brain injury, oxidative stress, inflammation and neuronal death from cyclophosphamide (in combination with Scenedesmus obliquus⬖), cisplatin (alone or in combination with DMFM)1,2,3 and doxorubicin (where in combination with curcumin⬖ prevented “chemobrain”).
•Prevent neural cell damage and death from a variety of metals such as lead (alone or in combination with thymoquinone⬖)1,2,3 aluminum (alone or in combination with GSK-3β, 3MA or dantrolene)1,2,3,4 cadmium, mercury (in combination with melatonin⬖ or curcumin⬖),1,2 the toxic form of manganese (alone or in combination with NAC or PAS-Na or a FTO inhibitor),1,2,3,4 toxic doses of lithium (in combination with curcumin⬖) along with arsenic (in combination with 3-MA), zinc nanoparticles (in combination with quercetin⬖) cobalt chloride (in combination with curcumin⬖) and fluoride (in combination with M3OMG), and thioacetamide.
Note: neuroprotective effects from DMSO in those studies included reductions in oxidative stress, neuronal cell death, calcium dysregulation, intracellular calcium release, birth defects, and histopathological brain damage.
•Protect animals from organophosphates, including otherwise lethal doses of nerve gas1,2,3,4,5,6 (or to enhance the efficacy of antidotes and reduce brain damage1,2,3) and to treat snakebites and their associated swelling in humans, cats, horses and dogs.1,2,3,4,5,6 Similarly, in two horses swarmed by African bees, IV DMSO as part of a combination protocol was able to reverse the severe neurological impairment created by the bee venom within five hours.
•In mice and rats, oxidative stress and neurotoxicity (e.g., in the hippocampus) from a variety of agents has been counteracted by DMSO in combination with another therapeutic agent: ethanol (nimodipine, DAPT or MSM),1,2,3 methamphetamine (curcumin⬖) mold aflatoxin (in combination with extracts of Chelidonium majus⬖ or artichokes⬖),1,2 liquid petroleum gas poisoning (a p38MAPK inhibitor), diethyl phthalate and bisphenol S (vanillic acid⬖), thrombin (estrogen), trimethyltin (carvacrol⬖), tunicamycin (4-PBA) chlorpyrifos (niosomal hesperidin⬖ or taxifolin⬖), calyculin A (melatonin⬖) fipronil (malvidin hydrochloride⬖), thapsigargin (Activin A). Likewise, melatonin⬖ mitigated PBDE-47 (fire retardant) neurotoxicity in PC12 cells.
Note: high-dose ivermectin causes neurotoxicity, limiting its use at higher doses. In one reported case, IV DMSO facilitated a full neurologic recovery in a comatose dog that had ingested a toxic dose of ivermectin paste.
Likewise DMSO also protects cells from a variety of harmful non-chemical stressors by:
•Protecting cells (including in a prophylactic manner) from being damaged by (often otherwise fatal) radiation exposures.1,2,3,4,5 For example, DMSO prevented X-ray and gamma ray DNA damage to hamster ovary cells, fruit flies and cerebral organoids (e.g., by accelerating DNA repair),1,2,3,4 and to prevent the harmful (bystander) signals irradiated cells emit in their vicinity from damaging non-radiated cells along with protecting certain bacteria from x-ray exposure.1,2,3 Likewise, DMSO has been repeatedly shown to reduce chromosome damage from radiation1,2 and prevent radiation from creating harmful free radicals. As such, DMSO has been shown to protect animals (e.g., mice, rabbits, dogs and monkeys) from often otherwise lethal doses of radiation, and prevent radiation tissue damage (e.g., to the bone marrow, intestinal lining, stem cells, eyes or skin),1,2,3,4,5,6,7 and, due to it preventing radiation damage in non-cancerous cells, DMSO has been extensively used as complementary cancer treatment.
Note: DMSO has been combined with many other substances to protect animals from radiation damage such as astragaloside-IV⬖ (preventing neuronal senescence), rapamycin (repeatedly preventing X-ray induced malformations of cortical development in rat offspring)1,2 thymoquinone⬖ (reducing brain peroxynitrite) or a glycogen synthase kinase-3β inhibitor (preventing brain tissue necrosis).
•Preserving the function of nerve fibers exposed to UV radiation.
•Treating a wide variety of burns (detailed here) and protecting the brain from heat damage (along with the previously mentioned cold injuries).
•Protecting cells from osmotic stress and dehydration (and in combination with nimodipine, protect neural cells from osmotic shock while inducing neurite growth).
•Protecting glial cells from being destroyed by sonic disruption via an ultrasonic vibrator (78% vs. 13% survival), and in conjunction with a TRPV4 antagonist, protect hippocampal neurons and microglia from infrasound-induced (16Hz/130dB) apoptosis.
•Preventing the dramatic increase in germ cell death, lifespan shortening, and oxidative stress caused by strong static magnetic fields and likewise preventing similar harm from continual exposure to electrically generated air ions.
•In combination with curcumin,⬖ protect fetal brain, kidney, and liver from damage caused by low-frequency electromagnetic field (EMF) exposure during pregnancy.
DMSO Safety
Every substance has an effective dose (how much elicits a therapeutic effect) and a toxic dose (how much elicits a harmful effect) with the balance between these two often deciding the value of a therapy (e.g., mercury partially treated syphilis but for centuries caused far more harm than good due to its severe neurotoxicity, and while often effective, certain modern therapies such as chemotherapy cause a lot of issues due to their toxic doses being so close to the therapeutic doses they are routinely prescribed for). Further complicating this, there is often no “correct” dose for everyone, so standardized ones are chosen which work for the majority of the population (which leads to sensitive patients those doses are too high for being routinely injured and then gaslighted by the medical system).
For this reason, I try to utilize therapies with a very wide therapeutic window (meaning the effective dose is much lower than the toxic one), but even in those cases, I still sometimes encounter patients who react to these “safe” doses and need much lower ones.
In the case of DMSO, what has been striking to me is how wide its therapeutic window is (demonstrated by the fact it has an extremely high, “practically non-toxic” LD50 and that rather than harm cells, it will protect them from a wide range of otherwise lethal stressors). Given this and how rapidly DMSO distributes and dilutes in the body, outside of animal experiments where large amounts of DMSO are injected, it is extremely unlikely DMSO can reach a toxic dose (so even in a human safety study where extreme doses were used for a prolonged period, no issues occurred).
That said, with (clean) DMSO, the following issues do exist:
Excessive DMSO commonly creates temporary itching and irritation on the skin. This can be avoided by using lower topical concentrations or a natural agent which prevents DMSO’s irritation—but nonetheless still routinely happens due to users using excessive doses.
In some individuals, DMSO will create an unpleasant odor (which often can be addressed through one of the methods detailed here).
A small number of people (one estimate pegged it at 1 in 2000) are allergic to DMSO. For this reason, it is advised to use topical DMSO first, ascertain if you are having an allergic reaction (rather than a typical skin irritation) and if so, back off, and absolutely avoid systemic applications (e.g., oral).
DMSO can bring toxic substances into the body, and in the early days of DMSO, there were rare reports of people becoming ill for a few days after having both DMSO and (now banned) pesticides contact their skin (along with one person who had a nicotine overdose). For this reason, it is generally advised to always wash your skin (with water) after applying DMSO (that has had time to dry), and in modern times, I have not come across reports of this being an issue.
DMSO will effectively transport allergens into the skin (leading to it being periodically used to patch test allergens). As some people are allergic to seemingly safe natural substances, this can occasionally cause issues (e.g., in the one report I received, hives followed a DMSO arnica preparation—as a surprising number of people are allergic to arnica).
All anti-coagulants carry the risk of excessive bleeding. Since DMSO uses a different method of anti-coagulation, this risk is much smaller, but still possible (e.g., three readers shared it seemed like their nosebleeds increased, and I’ve found one serious reaction in a case report).
One of DMSO’s greatest uses is it potentiating medications, but this also means it can increase their toxicity (even though, as the previous section shows, DMSO frequently counteracts toxicity). This has primarily been observed with alcohol, barbiturates, and to a lesser extent benzodiazepines (e.g., one cat study found adding diazepam to harmless IV DMSO caused fatal hypotension and ventricular fibrillation) but modern reports also exist of it increasing the frequency of side effects from more toxic medications (e.g., chemotherapy and fluoroquinolones). A major question is if this is also an issue with anticoagulants, as limited data and a lack of adverse reports suggest it is not, but simultaneously, due to the theoretical risk, we always advise patients to carefully monitor their blood coagulation (which is typically done for anticoagulant regimens). For all of these reasons, it is typically advised to take DMSO at least two hours away from pharmaceutical medications as this minimizes the potential for adverse potentiation.
Note: the issues with DMSO and alcohol are discussed in more detail here.In sensitive patients, temporary reactions to DMSO (e.g., headaches) can occur with excessive doses. I have also received two reports of extraordinarily sensitive patients (due to longstanding toxicity burdens) who became worse after DMSO due to it mobilizing stored toxins.
Over weeks, inhaling high doses of DMSO was found to harm rabbits, so for this reason, DMSO researchers (except for one successful ARDS study) avoided studying nebulized DMSO. Modern DMSO users, however, made the logical extrapolation to start nebulizing DMSO, and found significant benefit from it (e.g., remarkable results with COPD). Those who investigated this concluded a theoretical risk (they’d never observed) existed of nebulized DMSO neutralizing surfactant and collapsing the lung, so they cautioned against higher doses (which will also leach plastic from most nebulizers). Presently, one person (patient of a colleague) has had this happen to them (who due to their body type was already at high risk of a pneumothorax), but outside of that, a lot of people have successfully nebulized DMSO at much higher doses than we’d use without issue.
Similar concerns also existed with pregnancy as after injecting high doses of DMSO into or near fetuses was seen to cause developmental damage, very little research was conducted in this area (despite those doses being impossible to reach with standard DMSO uses). Fortunately, large numbers of pregnant and breast feeding parents having used DMSO without issue.
Note: in a future article I will compile all the research which has been done. Presently, the most definitive (but still not definitive) study showing DMSO safety in pregnant mothers is this one.When cells are exposed to high concentrations of DMSO for prolonged periods (which are impossible to reach in the body), cellular and microcellular injury will occur. Of note, the toxic DMSO thresholds for cancer cells are much lower than normal cells, which is likely one reason why DMSO is an excellent tool for cancer.
Because of the previous, virtually every study I’ve read which utilized DMSO did not report adverse effects from DMSO, serious adverse reports from DMSO are extraordinarily rare, and in almost all instances, those resulted from IV DMSO which was given in conjunction with an embolization agent or stem cells (whereas the much rarer ones from IV DMSO alone typically reversed once the infusion stopped1,2). As some of these were quite concerning, and did not match what we’d ever seen from IV DMSO (such as in this comprehensive safety study in monkeys or what I’d seen reported by countless practitioners using IV DMSO), I spent a while looking into this and concluded:
•The liquid embolic agents have independent toxicity and may sometimes travel and accidentally obstruct other parts of the circulation (as many of the reports seem to indicate this had happened, and once I checked, product warning labels acknowledged this).
•Many of the patients who receive IV DMSO stem cells are extremely fragile cancer patients (who went through high dose chemo), and hence are more likely to react to therapies, especially a higher doses. Likewise, one study found while dogs generally did not have issues with DMSO, those with chronic kidney disease did from higher IV doses.
•Due to DMSO’s safety, very high IV doses are used. These are often sufficient to create an osmotic shock which will rupture the weakest membranes (e.g., hemolysis is a common complication of higher IV DMSO doses, and in this horse study, was seen alongside significant—but temporary—symptoms when 40% DMSO at ten times the normal dose was rapidly infused into horses).
•The concentrations used for the previous applications are sufficient to leach phthalates from IV tubing. Phalates in turn can cause heart arrhythmias and one DMSO doctor found this was an issue with DMSO injected into the bladder until he switched to different tubing.
•Since DMSO increases parasympathetic activity through cholinesterase inhibition, it can slow the heart rate, particularly if a large amount of it suddenly reaches the heart, so while it typically does not alter cardiac rhythm and has been observed to normalize the reactivity of the autonomic nervous system, at high doses it can be arrhythmic (e.g., a 1-3% DMSO slightly increased the heart rate,1,2 while 6-10% significantly decreased it but could be reversed with atropine1,2).
•While practitioners (in recent days and throughout the DMSO literature) using much higher IV DMSO doses than we do do not appear to have run into issues, significant care in identifying appropriate IV DMSO dosing is likely warranted.
Lastly, due to DMSO’s widely recognized safety and negligible toxicity and ability to rapidly transport substances throughout the body, it is often used as an “inert” ingredient to deliver other pharmaceuticals and as a solvent or vehicle to facilitate evaluating the biological effects of large numbers of substances (as without being dissolved or transported, it is often impossible to test them).
Note: in addition to testing therapeutic effects, DMSO will also be used to deliver a harmful agent to trigger a disease1,2,3,4,5,6,7,8,9,10,11,12 (which makes it very time consuming to filter out therapeutically relevant DMSO studies) or to evaluate a therapy’s mechanism of action by seeing if the specific inhibitor DMSO delivers blocks the therapy’s efficacy (e.g., many Chinese acupuncture studies like this exist1,2,3,4,5,6,7).
As similar benefits are seen with many different therapies combined with DMSO (that DMSO alone would cause), this has led me to conclude:
•The toxicity of many toxins has been underestimated due to DMSO’s protective effects counteracting them (something also recognized by a few neurology researchers).
•A key reason benefits seen in pre-clinical studies do not appear in clinical ones (an extraordinarily common problem in scientific research) is because DMSO is no longer being used—particularly since I occasionally find studies where DMSO alone, rather than simply being the control, is also tested against saline, and in a significant number of those DMSO alone had a therapeutic effect.
Lastly, in writing this article, I have tried to condense thousands of pertinent studies into something feasible for everyone to read, while also ensuring that this forgotten literature remains available to researchers and authors wishing to advance this work. In that, I made the decision to include the combination studies, both because they illustrate the common benefits DMSO provides with these agents across a vast swath of neurological conditions and because combination can often enhance the efficacy of DMSO (detailed extensively here), hence providing additional ideas for individuals struggling with the neurodegenerative diseases covered here. As such, anytime an agent with a therapeutic effect is mentioned in this article assume that it was combined with DMSO. Additionally, if that agent was a natural therapy (e.g., herb, nutraceutical or botanical extract), to help you, as mentioned before, I have marked it with a ⬖.
Causes of Disease
When trying to understand a disease, two different lenses exist for interpreting it. One, the (favored) reductionist perspective tries to break it down to its tiniest parts, and through understanding them understand the disease. The other, the holistic one, sees the specific disease as a gear in a much larger system, and tries to see what systemic process is giving rise to the issues at hand.
Since Descartes’ seminal work on reasoning in 1637 (~400 years ago), our culture has embraced the reductionist model and through it, created countless scientific innovations which have transformed society, such as numerous medical innovations that identified the discrete cause of a life-threatening condition and provided a cure so it was no longer fatal.
Unfortunately, while reductionistic approaches are often excellent for acute life-threatening illnesses, they often only identify the downstream concrete effects of the illness rather than the upstream process which gave rise to the illness (hence making the therapies chosen typically be symptom managing ones rather than curative). Because of this, modern medicine is often characterized as being “excellent for emergencies but terrible for chronic illness.” Likewise, a longstanding joke with neurology is that neurologists are excellent at diagnosing neurological diseases, but not very good at actually treating them (although recently there has been some progress on the therapeutic end).
Note: alternatively, one can argue our biochemistry-focused form of medicine (which tries to identify a specific molecular target for each disease) exists because this allows an almost infinite number of patentable therapies to be made for each illness, whereas were systemic remedies to be utilized that could treat a myriad of illnesses (e.g., umbrella therapies or ones based on biophysics), it would no longer be possible to have a lucrative business model which patents each disease.
In contrast, I see many illnesses as being a manifestation of an underlying disease process within the body, and in many cases, believe the specific disease that arises is largely a product of where that disease process landed in the individual’s body (e.g., it was very common that COVID-19 vaccine injuries affected a previously weakened or injured area of the body, which is part of why the condition had so many different symptoms).
Unfortunately, while this perspective is often necessary to solve an illness, it is diametrically opposed not only to how our society teaches us to think, but also the human ego, as reductionist frameworks offer the comforting illusion of certainty and control, whereas holistic perspectives require us to tolerate ambiguity and unpredictability so we can see beyond the parts and grasp the broader whole—and unfortunately, the human ego will go to great lengths to feel like it is in control.
The Sequence of Disease
Over the years, I have noticed a recurring pattern that characterizes many of the diseases I encounter:
Something shocks the system, or a recurring issue eventually affects the body to an extent which exceeds its compensatory capacity.
The body (or a part of it) enters a state of shock and partially or fully shuts down.
The natural healing capacity of the body is unable to resolve this shutdown, and the issue becomes chronic.
The shutdown causes other things in the body connected to it to go haywire and creates additional issues.
Because of this, my approach frequently is to:
First identify where the actual issue is and the underlying issue that precipitated it.
Then treat the underlying issue which caused the problem.
See if that resolves the shutdown, and if not provide a regenerative therapy which wakes the tissue back up (which I discussed extensively in the cell danger response series).
For any problems that remain, treat the underlying issue which predisposed that area to being affected by the systemic process.
See what issues remain in the other parts of the body which were connected to the core problem and deal with those.
Note: in other cases, the situation is much simpler and I just focus on a therapy for where the actual issue is.
Because of this framework, I’ve put a lot of thought into what creates the shocks that initially shut the system down (e.g., an infection, prolonged stress, poor sleep, significant injury or tissue compression) and tried to discern why some people’s bodies can quickly shrug off those insults and the damage they create, while in others they become lodged and quickly become permanent.
From this, I’ve gradually come to the perspective that circulation is key, and that once circulation shuts down, areas of the body not only become “shocked,” but the body loses its inherent ability to reassert a state of health following the shocks it encounters. As such, I see many diseases and disease processes (e.g., inflammation) not as independent entities, but rather as consequences of poor circulation (and likewise recognize that the underlying reason why many different disease processes create similar symptoms is because they all impair circulation).
Note: within many schools of natural healing, nutritional deficiencies are identified as a root cause of illness to be treated with sufficient supplementation. My own experience (mirrored in some studies) has been that those illnesses often also resolve when circulation is restored to the affected area. Put differently, while raising the nutrient levels in the blood that reaches the area could solve the issue, those nutrients could also be obtained in sufficient amounts by increasing the amount of blood which reaches the area.
My focus on circulation in part results from how often I see it quickly produce dramatic effects for patients, in part because of how often I now identify pertinent circulatory obstructions, and because the individuals who pioneered this perspective provided one of the most illuminating models of disease I’d come across. Briefly:
•Building on work that came before him, in the 1940s to 1960s, Melvin Knisely elucidated that “blood sludging” (blood cells clumping together) underlay many illnesses, particularly hospitalizing ones as this generally reduced blood flow and eliminated microcirculation in vessels the clumped blood cells were too large to fit through. Key discoveries included burns, blood infections like malaria and cancer causing significant blood sludging (which then systemically affected the body) a few therapies (e.g., low molecular weight dextran and hydroxychloroquine) alleviating sludging, and by using a microscope to view vessels in the eye, it was possible to non-invasively assess how “sludged” the blood throughout the body was (whereas while sludging could be assessed in blood taken out of the body with live blood cell analysis or the ESR rate, it was not as accurate because the clumping behavior of blood always changed once it left the body).
•In Chinese medicine, numerous “disease patterns” exist to explain what is causing a specific illness. One of these, “blood stasis” (which I still need to write an article on) perfectly matches blood sludging, and interestingly, after the mass adoption of the smallpox vaccine, blood stasis more and more came to be seen as the primary cause of most illness. Notably, many diagnostic signs have been developed by Chinese medicine for blood stasis which have significant value in identifying “blood sludging.”
•While Knisely could tell blood sludging was a core cause of illness and that specific things triggered it (e.g., excessive heat or cold), he could not determine why it occurred and postulated it might be due to a sticky coating on the cells (not found in normal blood) that was tentatively identified as a protein.
•In the 1960s, Thomas Riddick, an engineer and chemist who regularly worked with colloidal solutions to thicken or thin them (e.g., clays need to be thinned to flow through pipes while sewage needs to be thickened so its waste matter clumps together and settles to the bottom) concluded his heart issues (which at the time were “incurable”) might be due to his blood being “too thick” and tried using the same agents he used to disperse industrial colloids on his body—which worked. This led to him concluding the primary variable he adjusted, zeta potential (the electrical repulsion between colloidal particles which allows them to resist agglomerating forces in a liquid system pushing them together) might underlie many different diseases and was the factor responsible for the blood sludging observed by Knisely. As such, he extensively studied it (e.g., with microscopes aimed at the eyes that filtered the heat from his incandescent bulb so exposed blood would not begin to sludge) and amongst other things concluded aluminum was extremely dangerous because its strong positive charge made it the ion most capable of disrupting zeta potential, that people with poor zeta potential were at high risk of heart attacks, and that bacterial and viral infections would consistently worsen the zeta potential of the body (hence making them most severely afflict the elderly as zeta potential worsens with age).
•A doctor with an incurable heart condition discovered Riddick’s work, and after it fixed his heart issues, discovered that with his patients restoring zeta potential was miraculous for a few other diseases including dementia.
•Canadian neurologist Andrew Moulden realized that he frequently saw children develop clinical signs of strokes after vaccination, and that more severe signs correlated to developmental disability following vaccination (mirroring a century of published case reports of cranial nerve issues accompanying vaccine encephalitis and those same deficits later being routinely observed in autistic children). Moulden then concluded that vaccines were causing microstrokes throughout the body due to zeta potential-disrupting agents in vaccines (e.g., aluminum) clumping blood cells together and because during an inflammatory response, larger white blood cells will obstruct the microcirculation (all of which was too small to detect with radiologic imaging and is a major reason why diagnostic tests cannot identify many chronic neurological conditions). He also concluded that the characteristic microstrokes he saw resulted from them being in parts of the brain with weaker blood supplies, and they hence served as indicators brain damage was also silently occurring in other parts of the brain being affected by these microstrokes. Finally, like those before him, he highlighted this could be caused by other things like infections, but emphasized it was a far more frequent problem with vaccination.
•Numerous doctors (myself included) independently realized that this process likely affected every fluid in the body as they are all colloids, and that many of the conditions ascribed to blood sludging (e.g., Chinese medicine links blood stasis to autoimmunity) likely resulted from obstructions in other fluids like the lymphatics.
•In December 2019, based on reports on anonymous message boards online, I became very concerned COVID-19 (SARS-CoV-2) would turn into a global catastrophe (in part because of how it behaved, and in part because every authority downplayed it, whereas typically far more minor and relatively inconsequential pandemics would be hyped up to an absurd degree). As such, from the start, I corresponded with everyone I knew treating the disease, and quickly noticed it had some very odd characteristics suggesting significant zeta potential disruptions throughout the body. As colleagues who had treated SARS-CoV-1 did not notice those features of the disease, I hypothesized there was likely a protein on the outside of the virus which carried a very strong charge density not present in SARS-CoV-1, and after teaching myself how to do the analysis, realized the spike protein fit the bill.
As such, particularly after the vaccine hit the market, my interest in understanding zeta potential greatly increased and a key goal of this newsletter has been to empower people to treat zeta potential (which is essentially done by eliminating strong positive ions and supplementing with strong negative ions) as it transforms so many different areas of medicine and health.
Note: the zeta potential topic (along with supporting references) is discussed in much greater detail here.
However, I also must disclose I do not believe zeta potential is the only factor which causes blood sludging; rather I’ve focused on it because it is simply the easiest one to understand and rapidly treat (e.g., I believe there is a great deal we still do not understand how blood and fluids behave in the body—evidenced by things like forgotten Russian research which shows blood travels through the body in spiraling vortexes the heart directs so vascular resistance is reduced and specific types of blood can arrive where they are needed).
DMSO and Neurological Disorders
DMSO has many qualities which allow it to treat a wide variety of diseases including:
•It increasing circulation.
•It accelerating the healing of injured tissue (which I currently believe results from it improving circulation and it stabilizing the gels needed for the initial healing process).
•It awakening dormant cells that are trapped in the cell danger response.
•It being a potent antioxidant and anti-inflammatory agent.
•It increasing parasympathetic tone (due to it being an acetylcholine esterase inhibitor) and it sedating dysfunctional neural circuits (allowing them to reset).
•It effectively reducing pain (in part by it blocking pain transmission and it relaxing the musculature).
•It protecting cells and tissue from a wide variety of injurious and lethal stressors.
•It being a potent delivery system for other therapeutic substances that are mixed with it (particularly in topical applications).
Of these, I believe the first three (circulatory improvement, tissue regeneration, and resetting the cell danger response—and possibly DMSO’s anti-inflammatory and anti-oxidant properties) are particularly important for neurological disorders (which DMSO has long been recognized for treating) as:
•Nervous system tissue has the highest energy demand in the body and is the most sensitive to its blood supply being reduced (e.g., functions of the nervous system will often immediately “turn off” once their blood supply is interrupted).
•Nervous system tissue is particularly vulnerable to interruptions in blood supply, and once this occurs, will often be stuck in a dormant “penumbra” state like the cell danger response (after which the tissue eventually dies). Furthermore, the brain and spinal cord are among the tissues most resistant to healing and regeneration in the entire body.
•Because so much of life depends upon a functioning nervous system, partial losses of function due to either of the previous create immediate noticeable consequences for the individual (whereas partial losses of function in the internal organs may not even be noticed without lab work).
Given all of this, I assumed that DMSO had to improve zeta potential, as while it has many proven anti-clotting properties (discussed here), many of the changes it created were identical to what would result from an improvement of microperfusion via a spacing out of red blood cells.
However, when I reviewed the literature, I discovered DMSO (due to it carrying a neutral charge) does not improve red blood cell zeta potential, and if anything, slightly worsens it. Likewise, DMSO is inherently viscous (thick), increases the viscosity of water by structuring it1,2,3 (which can be seen when the two mix together).
Note: this exothermic structuring is why DMSO will create heat when it mixes with water.
However, DMSO formulations have low viscosity,1,2,3,4,5,6,7 (or become negatively charged when prepared in DMSO), DMSO decreases the viscosity of bulk hydrophobic ions and most importantly, reduces blood cell aggregation (blood sludging) and blood viscosity which I believe is due to:
It behaving as a gel stabilizing (promoting) agent, which thereby forms water barriers between particles preventing them from aggregating (along with it making biomolecules like urea switch from opposing to supporting gel formation).
DMSO reducing the attractive forces between red blood cells Knisely identified (e.g., by neutralizing aggregating proteins), thereby allowing the existing zeta potential to become able to disperse the blood cells.
It likewise counteracting the agglomerating factors seen in pathologic states which otherwise cause blood cells to clump together. For example:
•DMSO (3%), by lowering blood cell viscosity (and increasing molecular mobility) completely prevented S. aureus from adhering to red blood cells (which if adhered would then cause blood cells to clump together).
•When DMSO was mixed with LPS (to model sepsis), rather than increase viscosity (which is a key issue in sepsis), blood viscosity decreased, and further decreased once resveratrol⬖ was also added.•In cancer, DMSO prevented the reduction in zeta potential (and mobility) which would otherwise occur in macrophages.
•Prevented positive ions from disrupting the zeta potential of negatively charged laponite.
•When exposed to radiation (gamma rays), lens proteins from the eye would aggregate and the viscosity would increase; DMSO prevented this.
•To establish a foothold in the lungs, bacteria which often colonize cystic fibrosis patients release cepacian, a polysaccharide which forms thick biofilms in the lungs’ (already thick) mucus, making it much harder for cystic fibrosis patients to breathe. DMSO in turn has been shown in laboratory studies to disrupt cepacian aggregates and halve their viscosity (which may partly explain why one physician shared their cystic fibrosis patient had life-changing improvements after DMSO). Furthermore, DMSO in combination with ivacaftor (a key medication used for CF) was found to reduce the viscosity of cigarette smoke-thickened lung mucus.
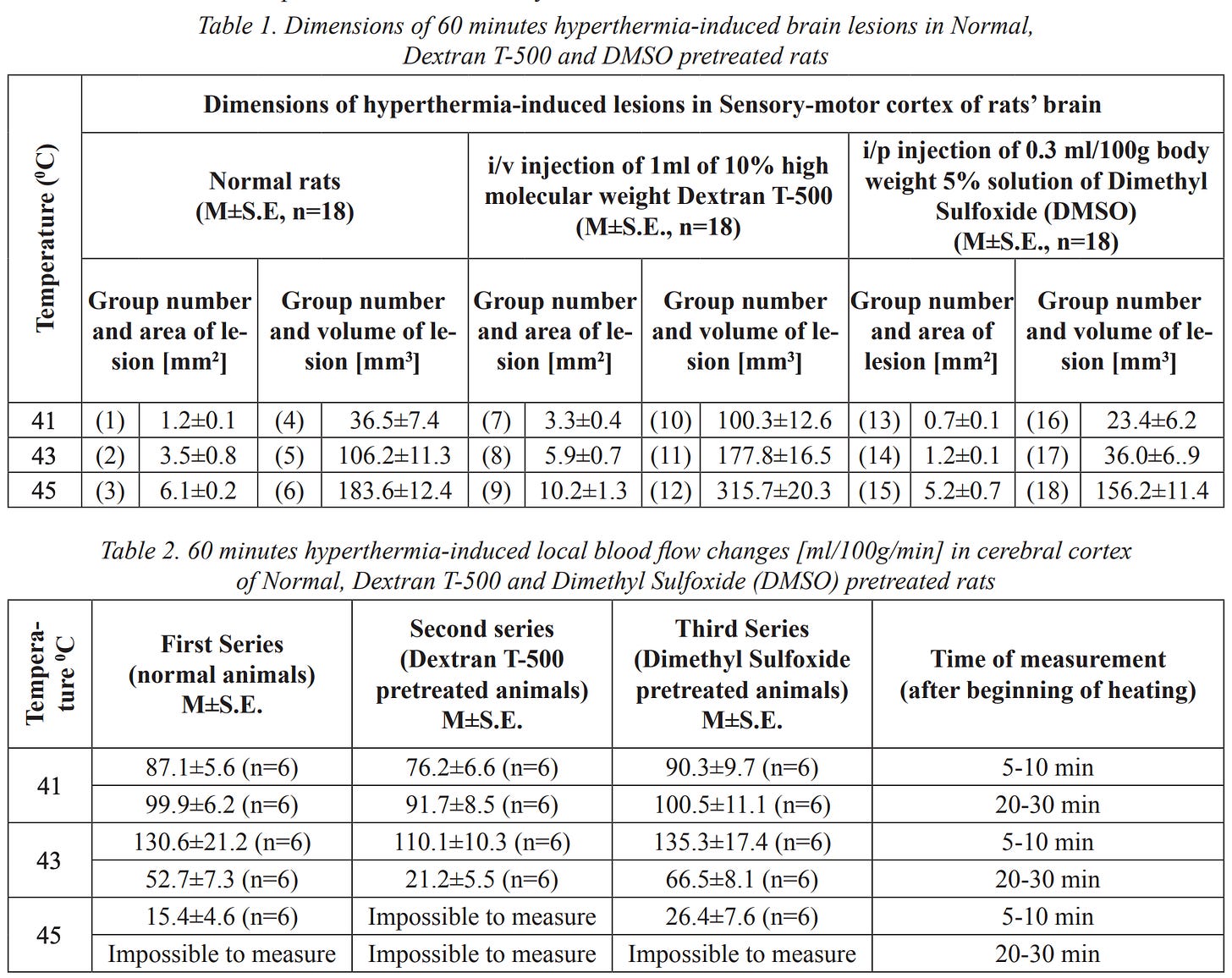
All of this was best demonstrated by a remarkable 2009 study1,2 by a team of Georgian researchers studying the effects of hyperthermia (a cancer treatment) on the brain as unlike the rest of the body, the brain and spinal cord are known to begin being injured by higher temperatures, with neurologic dysfunction beginning at 40-41°C and histological thermal damage (e.g., coagulative necrosis) occurring in primates after an hour of 44°C (which is why 43°C for 60 minutes is often considered the safety limit for hyperthermia treatment).
That study directly heated the CSF of rats, and then directly assessed the resulting blood flow changes in the brain and the lesions which followed, finding:
•Increasing heat caused increasing degrees of microclotting (mirroring Knisely and Riddick’s observations), corresponding losses of blood flow, and brain tissue damage from that loss of blood flow.
•DMSO counteracted all of these effects, preserving blood flow and brain tissue, while in contrast high molecular weight dextran (a substance Knisely used to induce blood sludging) worsened the sludging and brain damage—hence demonstrating why blood sludging can cause so many nervous system disorders, and why DMSO is able to antidote it.
Note: these results also make a strong case for using DMSO to mitigate the adverse effects of heat stroke and high fevers.
Additionally, DMSO has also been shown:
•In rat intestinal microcirculation, to reduce chemoattractant-induced leukocyte adherence (but not rolling velocity and flux), thereby counteracting the inflammatory microstroke-producing process Moulden had discovered of large white blood cells obstructing the smaller blood vessels.
• In microlymphatic vessels of intact rat mesentery to consistently stimulate phasic contractions, increasing the proportion of vessels exhibiting spontaneous phasic contractions from a baseline of 26–42% to 43–59%, while roughly doubling the contraction rate from ~11 to 25 per minute. As these contractions drive lymph flow, lymph velocity increased markedly — roughly doubling in 100% of vessels (via speckle-interferometry) and showing 40-100% increased movement in 60–64% of vessels (via direct microscopy) — thereby stimulating the drainage function of the lymph microcirculation. DMSO also completely removed the bacterial staphylococcal toxin’s lymphoconstrictive effects (which could progress to obliteration of microvessels), normalized lymphangion drainage, and attenuated the toxin’s overall lethal impact. Lastly, the Russian researchers who discovered this also found exposure greater than 15 minutes to 30% DMSO would induce lymphostasis in 20–40% of the vessels (concentrations that could never be reached in a patient using DMSO).1,2,3,4
•To increase lymphatic circulation across numerous contexts — including resolving lymphostasis in Kaposi’s sarcoma patients and in 115 patients with purulent wounds, facilitating lymphatic drainage (with electrical stimulation and hyaluronic acid), increasing renal microcirculation and lymph flow, increasing lymph flow in osteoarthritis, dose-dependently increasing lymphatic flow (as the solvent for intravenous Daflon), facilitating the growth of new lymphatic vessels (with 13-cis retinoic acid), and treating post-mastectomy lymphedema (per multiple guidelines, studies and reviews1,2,3). Additionally, a Russian detoxification patent first stimulated interstitial humoral transport and lymphatic drainage by applying DMSO combined with a proteolytic enzyme to the feet and then 30-60 minutes later, filtered the blood with plasmapheresis.
Note: DMSO is also used to directly deliver topical or injected therapies into the lymphatic system1,2,3,4,5,6,7,8 and to treat a wide range of lymphadenitis (e.g., from the BCG vaccine,1,2,3,4,5,6,7,8,9,10 tuberculosis,1,2 hemorrhagic erysipelas, Post-COVID MIS-C, tonsillitis, and bacterial infections which hospitalized 61 children1,2).
Lastly, a common side effect of IV DMSO is osmotic hemolysis, which predominantly affects aged blood cells (the spleen will eventually eliminate). Aged blood cells are well recognized to be much more prone to agglutinating and clumping (in part because red blood cells lose their negative charge with age) and as such, circulation will improve with their loss. This helps to explain Jack De La Torre’s observation with boluses of IV DMSO in brain injured patients:
Also, hematuria from red cell osmotic hemolysis was seen in all patients only after the initial loading dose of DMSO. Hematuria was seen to stabilize after subsequent doses of DMSO. The loading DMSO dose has no other consequences other than lowering the hematocrit about 25%, a temporary reaction that paradoxically lowers blood viscosity and vascular resistance while increasing cerebral blood flow.
DMSO and the Blood-Brain Barrier
In tandem with increasing circulation, DMSO rapidly spreads through the body, and within 5 minutes of going on the skin it can be found in the blood, within 30 minutes can be found throughout the organs, and within an hour, the bones—but simultaneously, does not accumulate within the body after prolonged use (e.g., 85% is excreted unchanged in the urine within 24 hours) and virtually none remains by a week after administration.1,2,3
Additionally, studies in mice and rats have shown that DMSO at 10–15% concentrations reversibly opens the blood-brain barrier (BBB), allowing proteins like horseradish peroxidase (HRP), many drugs including pemoline, ketoconazole (with brain concentrations increasing 9-fold), or the Parkinson’s medication L-dopa (increasing dopamine levels in the tubero-infundibular and neostriatal areas and further potentiated when combined with carbidopa), drug carrying lysosomes, and amino acids like tyrosine to reach brain tissue in higher amounts than without DMSO.1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20
DMSO also increased the transfer of amino acids across the subarachnoid space into underlying cortical tissue by approximately 57%, and in neonatal chicks, IV DMSO increased brain concentrations of adrenaline and noradrenaline by approximately 35–39% and intensified their central effects.1,2 Similarly, in dogs, IV DMSO at escalating infusion rates reached a CSF concentration roughly half the corresponding plasma concentration, confirming a (slower) penetration across the BBB into the CNS. This ability to facilitate drug delivery to the brain underpins DMSO’s therapeutic potential for neurological disorders (e.g., Parkinson’s) and has led to DMSO being grouped with mannitol as a clinical agent for enhancing brain drug delivery. Likewise DMSO has been incorporated into brain-targeting nanoparticle formulations, such as glucose-mediated poloxamer micelles, which showed significantly higher transport across a BBB model than ordinary micelles.1,2
Note: there are mixed results on DMSO temporarily opening the BBB (e.g., these eight studies found it did not,1,2,3,4,5,6,7,8 this one found that opening the BBB required 1% or greater DMSO, another found it had minimal effect on dopamine transport) these found the opening was also problematic,1,2 while in rats and rabbits DMSO infusions into the carotids did not harm the BBB, arteries or brain1,2 and a 1985 veterinary review likewise noted DMSO allows some substances but not others to cross the BBB.
In mouse MRI studies, DMSO accumulated at 1.5-fold higher concentrations in glioblastomas than in normal brain tissue with 2.2-fold longer washout, creating clear tumor "hotspots." Unlike toxic gadolinium contrast, DMSO freely crossed the intact blood-brain barrier, enabling visualization of low-grade tumors invisible to conventional MRI — and during chemotherapy, reductions in DMSO retention signaled treatment response earlier than volumetric MRI changes.1,2,3,4,5,6 DMSO has also been shown to enhance light penetration into brain tissue, improving optical diagnostic techniques relevant to certain neurologic disorders.1,2
DMSO and Psychiatric Conditions
Two of the most common complaints about psychiatry are that its (highly toxic) drugs do not treat the underlying illness—requiring lifelong symptom management—and that real biological issues (e.g., COVID vaccine injuries) are routinely misdiagnosed as primary psychiatric disorders. Both problems stem from the erroneous assumption that most psychiatric conditions originate purely in the mind, when in reality many have a clear biological (neurological) basis.
My perspective resulted from repeatedly observing psychiatric symptoms emerge after brain injuries and from seeing medical therapies that restore brain health also improve psychiatric issues. For example, one of my favorite therapies, ultraviolet blood irradiation works by improving circulation, reducing inflammation, and reawakening dormant cells—all of which DMSO also does. Because these processes underlie so many diseases, much in the same way a large body of literature supports UVBI’s efficacy across a wide range of conditions, including psychiatric ones, the same also holds true for DMSO (which I will show throughout this article).
In the process of unearthing every DMSO paper in existence, I uncovered a Russian team (at the Institute of Higher Nervous Activity and Neurophysiology of the Russian Academy of Sciences) which produced some of the best evidence I’ve come across for this theory.1,2,3,4,5,6,7,8
Briefly, in their effort to find a biologic cause of psychiatric conditions (which have a wide variety of seemingly unrelated causes), they discovered that the chronic stress which causes psychiatric disorders is accompanied by impaired circulation to the brain which sets off a variety of degenerative processes, especially once the individual’s ability to compensate for acute short-term stress is overwhelmed by chronic sustained stress.
To study this, they repeatedly induced neurosis in animals using prolonged stressors (white noise, light flashes, and electric shocks over 3+ weeks), then using biomicroscopy (cranial window) and hydrogen clearance, they directly measured blood flow in the territory of the middle cerebral artery, and finally, they directly examined the brains.
Note: neurosis (невроз) is an outdated psychiatric term. In this Soviet-era context, it describes a breakdown of higher nervous activity caused by chronic stress that overwhelms the person’s coping ability. Modern equivalents include Generalized Anxiety Disorder, mixed depressive and anxiety disorder (ICD-11), neurasthenia, and adjustment disorder with anxious or depressed mood.
From this they found:
•The brain normally receives 5–7 times higher blood flow per gram of tissue than most other organs due to its exceptional energy demands. Acute stress typically causes a short-term increase in cerebral blood flow, but prolonged chronic stress (leading to neurosis) produces a sustained decrease that persists 4–6 weeks after the stressor ends. This reduction causes circulatory hypoxia, elevated brain lactate, decreased caspase-3 and Na,K-ATPase activity, impaired mitochondrial respiration (including reduced succinate dehydrogenase and NADH dehydrogenase activity), and increased reactive oxygen species (ROS) production (and in animals unable to adapt by shifting from succinic acid⬖ to NADH oxidation, stress resistance is markedly reduced). The resulting hypoxia also increases cytochrome oxidase activity (by 35–40%) and triggers mitochondrial biogenesis, followed by ROS triggered lipid peroxidation (LPO).1,2,3,4
Note: the brain is particularly vulnerable to this cascade due to its exceptionally high metabolic rate and oxygen demand, as well as its high content of (oxidizable) polyunsaturated fatty acids in cell membranes.
•In the early stages of neurosis, acute stress, a nonspecific protective response, inhibits LPO, accumulates readily oxidized phospholipids, decreases cholesterol content, and increases superoxide-scavenging activity (partly from stress hormones acting as radical scavengers). With continued chronic stress, this protective phase is overwhelmed, leading to activation of free-radical lipid oxidation, progressive phospholipid depletion, cholesterol accumulation, and an increased in oxidized proteins. These biphasic membrane changes initially increase resistance to further peroxidation but ultimately render membranes more vulnerable as stress continues.1,2,3,4
Note: this biphasic pattern was also observed in women with dysmenorrhea (after 12 hours of pain, plasma levels of Schiff’s bases were reduced by almost two-thirds whereas after 12-24 hours it was nearly double from control values).
•The later adaptations to chronic stress are more specific and membrane and free-radical changes often show interhemispheric asymmetry varying by behavioral type. For example, in acute stress, animals with high emotional reactivity and emotional resonance shift toward balanced or right-dominant LPO (as do stress sensitive rats that excel with mazes), while low-reactivity animals have more LPO on the left (as do stress resistant rats that are not good with mazes).
•These molecular changes are accompanied by clear physiological disturbances: elevated and fluctuating systolic blood pressure, disruption and reduction of local cerebral blood flow, loss of functional specificity (equalization of blood flow across brain structures), and a behavioral shift toward passive-defensive behavior. As stress and sympathetic hyperactivity can cause these autonomic disturbances, the researchers concluded that the resulting restriction of cerebral blood flow contributed to many symptoms seen in neurosis.
Note: one of my favorite modalities (neural therapy) works by neutralizing autonomic disturbances and frequently produces rapid, dramatic responses in complex illnesses — which I believe relates to the pathological process described by the Russian researchers.
•They also found that local norepinephrine release within the lateral hypothalamus functions as part of a depressor system that helps normalize elevated blood pressure. During acute stress, the speed of return to baseline blood pressure depended on the strength of this local noradrenergic response in the lateral hypothalamus. As such, when this mechanism is impaired, repeated stress can lead to prolonged hemodynamic instability, which over time, can contribute to dysregulation of cerebral autoregulation and sustained reductions in cerebral blood flow (which then damages the hypothalamus, creating a downward spiral into chronic illness).
Note: most chronically elevated blood pressure has no known cause. This framework potentially explains a key unrecognized cause (along with another reason why restoring zeta potential will improve blood pressure, as doing so will restore blood flow to the hypothalamus). Additionally, I should note that certain holistic healers have reported significant success in treating excessive sympathetic activity by addressing hypothalamic function.
•Normal cerebral blood flow is approximately 50 ml/100 g/min; in chronic neurosis it falls below 30. Cerebral vessels also lose autoregulatory capacity: after bilateral carotid occlusion, normal animals show universal arterial dilation, whereas neurosis animals exhibit mixed arterial and venous responses (e.g., in arterioles 54% dilated, 21% constricted, 25% had no change) with frequent spastic contractions, bottle-shaped deformations, interrupted flow and perverted pial vascular reactions, resulting in a relative equalization of blood flow rate across all studied structures (indicating a loss of functional specificity—which I consider to have immense clinical significance as a few healing traditions associate this circulatory shift with approaching death).
Note: in many cases, cerebral hemodynamics never fully recover after the chronic stress period.
•These hemodynamic changes parallel the homogenization of EEG activity seen in neurosis. Biomicroscopy confirmed microstructural disturbances consistent with hypoxia, including perivascular and pericellular edema, tortuous vessels, dark neurons, acidophilic cells, microglial proliferation, and hippocampal damage (especially shrunken soma, altered nuclei, and corkscrew dendrites in CA3; 2.7–7.1% cell loss in CA1—approaching the threshold for cognitive impairment and dementia).
•The process selectively damages brain β-adrenoreceptors (which for about a week showed decreased receptor affinity that was compensated for by an increased receptor number, with the elevated receptor density persisting after three weeks), the sensorimotor cortex (layer V), and the hippocampus (in the pyramidal layer, particularly at the CA3 field).
•Three stages of the general adaptation syndrome were identified in the chronic emotional-painful stress model: (1) initial search for optimal functioning with residual visceral defects, fear-dominant behavior, and labile blood pressure, lasting a week; (2) partial autonomic stabilization but ongoing phospholipid depletion (“local wear”); (3) exhaustion with breakdown of autonomic regulation, LPO activation, and profound membrane disruption across neurons, glia, and synapses, contributing to the breakdown of higher nervous activity, which they described as “pathological adaptation with a high structural price.”
They then:
•Emphasized that an individual’s internal reaction to stressors rather than the stressor is pivotal (“...it does not matter what facts are reported to us — what is important is how we react to them; that is the main question”) and linked it to the observation that many illnesses resulting from chronic psychoemotional stress are characterized by autonomic (vascular) disorders, hypoxic states, and serious disturbances in metabolic processes, often manifesting as autonomic/vascular dysregulation, hypoxia, and metabolic disturbances.
Note: the non-English speaker who coined the medical concept of stress later stated he used the wrong word and meant to use the word strain (how a system deforms in response to stress).
•Noted that, while many individuals reach full neurosis, far more are in a pre-neurotic stage of significant strain without complete decompensation and would greatly benefit from therapeutic interventions early in their disease process.
•Highlighted that the effects of chronic stress they observed were similar to those seen after strokes, heart attacks, or traumatic brain injury, and in many cases, they successfully used the same therapies for both (e.g., panthenol⬖).
To address neurosis:
•The researchers first used agents with antihypoxic and antioxidant properties (e.g., carnosine,⬖ substance P, phenosan K, or synthetic phenolic antioxidants1,2) and found these interventions both prevented and effectively treated experimental neurosis in animals (whereas untreated animals consistently developed neurosis and showed poor recovery).
Note: other agents like panthenol⬖ only gave temporary improvements. Additionally, they also had significant success with alcohol (a hydroxyl scavenger), providing a novel explanation for why alcohol offers some relief from chronic depression. Notably, chronic stress decreased brain Na,K-ATPase activity (as detailed above), and a separate study on brain Na,K-ATPase found that while both DMSO and ethanol scavenge hydroxyl radicals, ethanol further destabilizes the enzyme whereas DMSO stabilizes it — potentially explaining why DMSO provides lasting benefit where alcohol only temporarily palliates (while in parallel, Riddick found alcohol increased blood sludging).
After testing multiple agents, the researchers achieved strong success with negative ion therapy (which has a pronounced antihypoxic effect). When present during acute stress (e.g., immobilization), negative ions completely prevented the pathologic brain changes in all animals — regardless of behavioral type — including preservation of oxidative enzyme activity in the sensorimotor cortex and normalization of behavioral and autonomic parameters (heart rate, blood pressure, and breathing). Similar protective effects were observed with succinic acid⬖ (30 mg/kg orally daily for 8 days), which they also found preserved orienting behavior after a heart attack. Notably, rats with an active behavioral type showed greater natural resistance to cerebral hypoxia, exhibiting faster increases in local cerebral blood flow and brain oxygen tension during stress.
Note: positive ions in the air have been extensively linked to psychiatric conditions. I believe this is because positive ions impair zeta potential and hence reduce cerebral microcirculation (whereas negative ions restore it).
They eventually had the greatest success by combining oral DMSO (a potent hydroxyl scavenger) with vitamin E⬖ (alpha-tocopherol), finding the efficacy of this combination exceeded them being given separately (e.g., for autonomic or behavioral issues). They attributed this to DMSO enhancing vitamin E⬖’s antioxidant capacity as DMSO could rapidly deliver it to cell membranes before it had lost its antioxidant capacity from reacting with other substances in the body (supported by it reducing free radical oxidation products, raising superoxide scavenging activity in the brain and blood serum, raising brain phospholipid content and normalizing brain cholesterol content). Finally, in 1999, they shared these results had begun being replicated in government sanctioned clinical trials at the Moscow Medical Academy.
While much could be said about their research, one of the key lessons I received was one of the clearest mechanistic explanations I’ve encountered for what adaptogens actually do (a term applied to many natural products) as the agents which effectively counteracted the entire stress process were explicitly characterized by the researchers as “adaptogens.”
Note: to compile the above summary and accurately represent their findings, I read through over 50 papers (many of which omitted key details) and did my best to integrate their findings with current physiologic science.
With this understanding (and a further exploration of the importance of circulatory drainage which will be discussed in the Multiple Sclerosis section), let’s now examine how these properties allow DMSO to affect a wide range of neurological and psychiatric disorders.
Parkinson’s Disease
Parkinson’s disease results from the progressive loss of dopamine-producing neurons in the substantia nigra. Research in this field was revolutionized in the early 1980s when recreational drug users who injected a badly synthesized synthetic heroin rapidly developed severe Parkinson’s-like symptoms due to it being contaminated with MPTP, an agent whose active metabolite (MPP+) specifically targeted those neurons, making it possible to reliably model Parkinson’s in laboratory animals. This was followed by the realization that one herbicide (paraquat) was very similar to MPP+, another pesticide (rotenone) also causing similar damage to neurons, a variety of pesticides being linked to a higher risk of Parkinson’s (such as organophosphates), and 6-OHDA also being able to reliably create Parkinson’s.
Note: one of the major challenges with glyphosate (Roundup) is that while it is toxic, the herbicides it largely replaced like paraquat are more toxic.
Numerous studies have shown that DMSO directly counteracts the neurotoxicity of these Parkinson’s-producing agents (e.g., in the organophosphate studies mentioned previously, DMSO repeatedly reduced mortality, accelerated organophosphate detoxification, and protected neuromuscular function). Most remarkably, a case-control study of young-onset Parkinson’s disease (63 cases, 68 controls) found that individuals with Parkinson’s were one tenth as likely to have been exposed to DMSO as normal controls, suggesting DMSO exposure is associated with a roughly 10-fold reduction in disease risk (and hence may be protective against it). In contrast, the same study found insecticide exposure increased risk nearly 6-fold, fumigated housing over 5-fold, and herbicide exposure over 3-fold — results consistent with the extensive epidemiological literature linking pesticide exposure to Parkinson’s.
Note: this study also found smoking was associated with reduced PD risk, a finding that aligns with decades of epidemiological evidence linking nicotine exposure to lower PD incidence, lending credibility to the study’s methodology.
DMSO has directly demonstrated neuroprotective effects in multiple Parkinson’s models. In animals, DMSO suppressed hydroxyl radical-induced nigrostriatal injury from MPTP,1,2,3,4 and in rotenone-induced Parkinson’s rats, DMSO improved hippocampal CA1 and CA3 neuron morphology, restoring pyramidal cells and Nissl bodies damaged by rotenone and normalizing their electrical activity. DMSO also protected astrocytes from MPP+-induced toxicity by reducing lipid peroxidation and metabolic impairment, protected glial glutamine synthetase from MPP+-induced hydroxyl radical damage, protected human SH-SY5Y neuroblastoma cells from 6-OHDA-induced cytotoxicity, and reduced both lipid peroxidation and protein carbonyl formation in rat brain homogenates from ferrous chloride or hydrogen peroxide, and separately reduced hydroxyl radical production during 6-OHDA autoxidation and the formation of hydroxylated dopamine products.1,2
Note: in one mouse study, intraperitoneal DMSO did not protect against MPTP-induced dopamine depletion, indicating its neuroprotective effects may depend on the route, timing, or dose of administration.
Interestingly, DMS (DMSO’s naturally occurring, odor-producing metabolite) at near-physiological concentrations also protected neurons against both 6-OHDA and MPP+-induced apoptosis, with this effect being dependent upon MsrA (the enzyme that converts DMS to DMSO), suggesting the endogenous DMS-DMSO cycle functions as part of the body’s natural antioxidant defense against dopaminergic neurodegeneration.1,2 This, in turn, raises an interesting conundrum as I have received a few reports of Parkinson’s patients who had dramatic responses to DMSO who then stopped due to the odor impeding sexual relations with their spouse, and my first thought was to recommend a low odor DMSO formulation (discussed here), but if DMS plays a key therapeutic role in Parkinson’s disease, that approach may not be viable.
Note: that study also found DMS protected against H₂O₂-induced lipid peroxidation and antimycin A generated superoxide production.
Additionally, DMSO reversed rotenone’s complete blockade of microtubule assembly from purified tubulin in vitro — a finding with direct relevance to Parkinson’s, as microtubule disruption impairs axonal transport and contributes to dopaminergic neuron death. Likewise, a Russian physical therapy monograph recommended topical DMSO novocaine compresses for neurological conditions including Parkinson’s, and a patent proposed DMSO as a transdermal enhancer for a botulinum toxin patch to treat the spasticity associated with Parkinson’s, cerebral palsy, dystonia, and multiple sclerosis.
Note: a large number of studies (which will be discussed later in this series) show DMSO stabilizes microtubules and likely accounts for some of its neuroprotective qualities.
A vast number of agents in combination with DMSO have also shown therapeutic benefit in Parkinson’s models.
Curcumin⬖ protected nigral dopaminergic neurons, reduced iNOS and glial activation, and upregulated neuroprotective pathways (IGF-1/Akt/FoxO3a).1,2
Paeoniflorin⬖ repeatedly reduced α-synuclein expression, decreased Lewy body formation, and protected dopaminergic neurons across multiple studies.1,2 It also inhibited microglial overactivation, increased BDNF and GDNF secretion, and promoted neural stem cell differentiation into dopaminergic neurons.1
Icariside II⬖ induced human amniotic mesenchymal stem cells to differentiate into dopaminergic neuron-like cells (optimal at 3–10 μmol/L via PI3K signaling). In another protocol DMSO helped differentiate iPSCs into dopaminergic progenitors for PD stem cell therapy.
Ginsenosides Rg1⬖ and Rg3⬖ both significantly attenuated dopaminergic neuron loss, neuroinflammation, and α-synuclein accumulation.1,2,3,4
Geniposide⬖ reduced α-synuclein levels and prevented dopaminergic neuron loss by modulating the miR-21/LAMP2A axis, while ginkgolide B⬖ similarly reduced α-synuclein expression via the related miR-207/LAMP2A pathway.1,2 Ambroxol increased β-glucocerebrosidase activity and reduced α-synuclein oligomer levels, restoring cell viability and mitochondrial function in dopaminergic neurons. Polyphenols⬖ reduced seeded α-synuclein aggregation via NRF2-mediated antioxidant responses. Carnosic acid⬖ attenuated 6-OHDA neurotoxicity by upregulating parkin and restoring proteasomal clearance of ubiquitinated proteins in cellular and animal PD models.
L-sulforaphane⬖ dissolved in DMSO activated the NRF2 pathway in Parkinson’s disease patient-derived cells, restoring their deficient glutathione levels — one of the only studies using actual patient cells.
Most uniquely, NAMI-A — a low-toxicity ruthenium-DMSO complex — inhibited α-synuclein aggregation and membrane interactions with submicromolar affinity, disassembled pre-formed fibrils, abolished α-synuclein cytotoxicity toward neuronal cells, and mitigated neurodegeneration and motor impairments in a rat Parkinson’s model, providing a novel basis for designing ruthenium-DMSO complexes that target α-synuclein-driven pathology through a mechanism distinct from organic agents.
In MPTP models, tanshinone IIA⬖ preserved approximately 75% of dopaminergic neurons while reducing microglial activation; tetramethylpyrazine⬖ prevented motor deficits and neuron loss via the Nrf2 pathway; 6-Hydroxy-1H-indazole protected 90-93% of dopaminergic neurons from death; baicalein⬖ dose-dependently reduced rotation behavior (a key indicator of motor impairment), neuroinflammation, and dopaminergic neuron apoptosis via Wnt/β-catenin; neferine⬖ improved mouse motor disorders and reduced neuroinflammation and α-synuclein in the substantia nigra; SB239063 (a p38 MAPK inhibitor) protected TH-positive neurons; NESS 0327 (a CB1 receptor antagonist) ameliorated motor deficits; novel c-Abl kinase inhibitors outperformed nilotinib in blocking MPP+-induced apoptosis; GW5074 prevented TH-positive neuron loss in mice genetically engineered to have PD. In nigrostriatal pathway injury mice, both ERK inhibition (U0126) and PDGFRα inhibition (AG1296) reduced glial activation and scarring, with U0126 also improving long-term neurobehavioral outcomes.
In LPS-induced PD mice, pazopanib protected dopaminergic neurons by suppressing TNF-α, PGE2, and IL-6 via MEK4-JNK-AP-1 signaling, while rapamycin reduced neuroinflammation by enhancing microglial lipid metabolism.
NBP (a Chinese stroke medication) rescued dopaminergic neurons by 30% and striatal dopamine terminals by 49%. Carvacrol⬖ (found in oregano and thyme oils) was neuroprotective via TRPC1 inhibition in dopaminergic neurons and TRPA1 activation in astrocytes. Dasatinib and resveratrol⬖ in combination improved learning, memory, motor coordination, and reduced anxiety. MOTS-c⬖ improved motor function, reversed TH-positive neuron loss, and activated the Nrf2/Keap1 antioxidant pathway in rotenone PD rats. Puerarin⬖ mitigated rotation behavior and upregulated DAT, VMAT2, and TH in rotenone PD rats. A caspase inhibitor reduced neuron loss and improved rotation behavior in 6-OHDA rats, though blocking apoptosis triggered compensatory glial necroptosis.
Shuimuheningfang⬖ improved motor and non-motor symptoms in 80 PD patients and reduced α-synuclein in model mice,1,2 while Compound Dihuang Granules⬖ (with a JNK inhibitor) reduced rotation behavior and protected dopaminergic neurons in 6-OHDA rats.
In C. elegans PD models, olive leaf extract⬖ strongly protected dopaminergic neurons from 6-OHDA toxicity (up to ~56% less degeneration), while oleuropein,⬖ oleanolic acid,⬖ tyrosol,⬖ 3-hydroxytyrosol,⬖ saffron,⬖ Polygonum multiflorum,⬖ and Ziziphus jujuba⬖ each also provided significant protection.
Additional agents showing neuroprotective effects in PD models include guaraná⬖ (against rotenone in SH-SY5Y cells), Antarctic krill oil⬖ (improved locomotor activity and dopaminergic neurons in zebrafish), lutein⬖ (dose-dependently improved cognitive and motor outcomes in rats), cytochalasin compounds from endophytic fungi (against MPP+), Erythrina velutina extract,⬖ rizonic acid,⬖ and xyloketal derivatives⬖ (against 6-OHDA or ROS-mediated damage), sodium butyrate⬖ (an HDAC inhibitor that epigenetically restored dopamine transporter and VMAT2 expression against rotenone and MPP+), allopregnanolone (promoted TH-positive cell regeneration via BDNF and CaMKIIδ3 against 6-OHDA), wedelolactone⬖ (upregulated the neuroprotective PD protein DJ-1/PARK7), dexmedetomidine (neuroprotective via ERK1/2-mediated histone acetylation), along with 7,8-dihydroxyflavone,⬖ cordycepin⬖ (against rotenone in PC12 cells), AMG9810 (a TRPV1 antagonist that reduced motor deficits but impaired cognition with chronic use), insulin with TLR4 inhibitor TAK242 (improved motor performance and normalized α-synuclein in 6-OHDA rats), catalpol⬖ (reduced α-synuclein and improved mitochondrial function against rotenone), genistein,⬖ Taohe Siwu decoction,⬖ Ligusticum chuanxiong compounds,⬖ and Nigella sativa fatty acids.⬖
Since paraquat and other herbicides are among the strongest environmental risk factors for Parkinson’s, it is also noteworthy that DMSO has been shown across multiple studies to scavenge the hydroxyl radicals generated by paraquat,1,2,3,4 including direct evidence from rats of DMSO intercepting paraquat-generated hydroxyl radicals via Fenton-like chemistry, and in bacterial biosensor assays, DMSO scavenges up to 96% of the superoxide radicals generated by paraquat. DMSO has also been shown to be directly neuroprotective against paraquat in cultured striatal cells, suppress paraquat-induced inflammatory signaling (e.g., IL-8 and neutrophil chemotactic activity), and protect DNA from paraquat-induced mutagenesis — providing a potential mechanistic explanation for the epidemiological finding that DMSO exposure is inversely associated with Parkinson’s risk. Additionally, myrtenol,⬖ andrographolide⬖ (via Nrf2/HO-1), VPA (an HDAC inhibitor), chymostatin, propofol and resveratrol⬖ each combined with DMSO to counteract paraquat-induced toxicity and oxidative stress across various tissue models.
Note: α-synuclein aggregation into toxic fibrils is a core driver of Parkinson’s neurodegeneration. One study found DMSO at 0.75-1.0%, especially when combined with ferric iron, promoted α-synuclein oligomer formation and cytotoxicity. However, when oral DMSO was tested in living mice (both normal and transgenic mice overexpressing human α-synuclein), no increase in α-synuclein aggregation, no neuronal loss, and no Parkinson’s-like pathology was detected. Likewise, DMSO injected directly into the substantia nigra has not been found to cause dopaminergic neuron loss, ubiquitinated protein accumulation, or behavioral deficits1,2 — suggesting that whatever pro-aggregant effect DMSO has on α-synuclein in isolated cell cultures (at much higher concentrations than it can reach clinically) does not translate to the living body.
In addition to the experimental evidence, I have received a few reports from readers and physicians who had success with DMSO. As my experience is primarily with IV DMSO (which I believe offers the greatest benefit), I wanted to share this entire sample which includes non-IV approaches to illustrate the difference between them.
One wife described what happened when her husband with Parkinson’s received an IV drip of mannitol and DMSO during stem cell therapy in Amsterdam: “He bounced down a flight of stairs without using the handrails, cut his own food for a week after, spoke clearly, opened cab doors.” They knew it wasn’t the stem cells, which would take months to show results.
The most detailed report came from a research scientist diagnosed with PD in 2018, who had already controlled his non-motor symptoms with sulforaphane⬖ (an Nrf2 activator) but still had the full range of motor symptoms. After systematically testing oral DMSO over several months, he found that at an optimal dose, bradykinesia was eliminated, pain and dystonia reduced by 80%, stiffness reduced by 50%, and energy levels were markedly higher. He observed that DMSO addressed motor symptoms where sulforaphane⬖ had not, suggesting DMSO was reaching the brain in ways sulforaphane⬖ could not — consistent with DMSO’s known ability to cross the blood-brain barrier. Notably, doses above his threshold reliably worsened tremor, stiffness, and sleep, but these effects fully reversed within two days of stopping.
A third reported that topical and oral DMSO initially helped her husband with Parkinson’s walk short distances, but the effect did not persist.
Additionally, I have also received a few reports of oral DMSO helping readers with Parkinson’s, but as they were in passing (verbally) I can’t offer any specifics on them.
Given all of this (and our own experiences with Parkinson’s), I believe DMSO has great potential for Parkinson’s disease — oral administration is likely to benefit patients, IV significantly more so — and that the best results will ultimately come from combining DMSO with a complementary neurotrophic agent. Currently, I have identified one very promising candidate for this purpose (along with a few other possibilities), but as the combination studies above demonstrate, there are likely many more waiting to be discovered.
Amyotrophic Lateral Sclerosis (ALS)
Amyotrophic lateral sclerosis is a progressive neurodegenerative disease in which the motor neurons that control voluntary movement gradually die, leading to increasing muscle weakness, paralysis, and typically death within 2–5 years of diagnosis. No cure exists, and the few FDA-approved drugs provide only modest survival benefits. However, as Todd’s story shows (particularly since he was willing to prove DMSO worked by repeatedly discontinuing it and then documenting the rapid worsening of symptoms that started), there is hope for ALS. In turn, there is also some research to corroborate it:
•In ALS model mice, long-term oral administration of 5% DMSO significantly increased mean survival time, reduced neurological scores, and improved motor performance (with the improvements being primarily functional rather than histological).1,2
•Low concentrations of DMSO were found to stabilize SOD1 protein conformation (SOD1 misfolding is a central cause of ALS). Additionally, 5-fluorouridine and epigallocatechin gallate⬖ (which is often combined with DMSO) also stabilized SOD1.
•A variety of agents in combination with DMSO have also shown therapeutic benefit in ALS mice. Chronic intraperitoneal resveratrol⬖ delayed disease onset, extended survival, and preserved nearly twice as many motor neurons. A GSK-3β inhibitor delayed disease onset and death, and partially preserved lumbar motor neurons. ASK1 inhibitors protected against motor neuron death and reduced glial activation. Rapamycin improved the neuroprotective mitochondrial fission/fusion balance. Lycopene⬖ dose-dependently reduced oxidative stress and reduced motor neuron apoptosis. Notably, carboxyamidotriazole potently inhibited inflammatory cytokines in vitro but did not significantly improve onset or survival compared to the DMSO vehicle control in vivo—potentially suggesting DMSO itself was already providing a comparable benefit.
Note: DMSO has also been combined with riluzole (one of the only ALS drugs) to treat a variety of other neurological conditions including neuropathic pain,1,2 light-induced retinal degeneration,1,2 hearing loss (where DMSO alone also was found to protect hearing and preserved cochlear neurons), and status epilepticus—all of which data shows DMSO treats. Additionally, DMSO has been used as a solvent to screen large numbers of compounds for use in ALS.
In addition to Todd’s remarkable response to topical and particularly IV DMSO, a few other reports suggest DMSO can benefit ALS and related conditions. One book recounts Stanley Jacob treating an ALS patient with DMSO, producing “instant, overnight and slightly delayed wonders of therapy” (after which their doctor forbade further treatment).
Note: I suspect this case may have been what first inspired a mentor to try IV DMSO for ALS.
Another reader reported that a colleague gave DMSO to her father with ALS and “was surprised at visible improvement in his condition.” Finally, a reader with cramping fasciculation syndrome (a condition that presents like early ALS but does not progress to it) described being driven to the point of planning suicide by the combination of chronic pain and severe sleep deprivation before discovering that oral DMSO dramatically improved the effectiveness of his other medications, allowing him to sleep through the night, largely eliminating his cramping and nerve pain, and giving him back the ability to hold down a job and watch his children grow up.
Note: our (limited) experience has been that IV DMSO halts the progression of ALS rather than reverses it. However, the stories I’ve received suggest some individuals have a considerably more dramatic response — whether due to inherent responsiveness or higher IV doses. One of my major unresolved questions is whether the post-COVID vaccine “atypical” ALS cases respond differently to DMSO than the pre-vaccine ALS we had previously encountered (which is where all of our experience comes from).
Huntington’s Disease
Huntington’s disease is a fatal genetic disorder characterized by progressive loss of motor control, cognitive decline, and psychiatric disturbances. It belongs to a family of nine neurodegenerative disorders (polyglutamine diseases) caused by misfolded proteins with abnormally long glutamine repeats, and a review of chemical chaperones for these conditions found that DMSO showed similar or superior suppression of polyglutamine-mediated toxicity compared to the other chemical chaperones tested (glycerol, TMAO). Likewise, in cell models of Machado-Joseph disease (another member of this family), DMSO stabilized mutant ataxin-3 protein folding, reducing aggregation, cytotoxicity, and cell death.
In the only study which directly tested DMSO against Huntington’s disease, DMSO (~1-4%) partially prevented cell death, increased cell viability, decreased aggregated huntingtin protein, and increased its soluble (non-toxic) form.
A variety of agents in combination with DMSO have also shown therapeutic benefit in Huntington’s models. In rats, inosine⬖ protected against Huntington’s-like symptoms by improving motor function, activating the neurotrophic BDNF/TrkB/ERK/CREB pathway, increasing striatal BDNF, and reducing oxidative stress, neuroinflammation, and striatal neuronal damage. FKBP5 inhibition reduced mutant huntingtin levels and increased autophagic clearance in both human Huntington’s stem cells and mouse models. In C. elegans Huntington’s models, olive leaf extract⬖ strongly protected neurons from polyglutamine-induced degeneration (up to 4-fold more intact neurons) and improved mechanosensory response, while 3-hydroxytyrosol⬖ and tyrosol⬖ reduced polyglutamine plaque number, protected neurons, and improved mechanosensory response. Hyptis species extracts⬖ also significantly improved locomotion and increased oxidative stress resistance in polyglutamine models.
Note: DMSO has also been used to screen a large number of potential therapeutics for Huntington’s disease and to investigate its underlying pathogenic mechanisms.
Alzheimer’s Disease
Alzheimer’s disease (AD), the most common form of dementia, is characterized by the extracellular accumulation of amyloid-β (Aβ) plaques (particularly in the hippocampus and cortex) and intracellular neurofibrillary tangles from hyperphosphorylated tau protein. It is thought to result from the damage those proteins are known to cause to brain tissue and as such virtually all research into the disease has revolved around removing amyloid.
Note: a strong case can be made amyloid proteins initially protect brain cells from stressors (e.g. toxins), supported by the fact all the amyloid eliminating drugs have failed (and often have significant side effects due to them priming the immune system to attack amyloid and hence create inflammation in the brain), along with the fact natural therapies which directly target the factors injuring brain tissue are currently the only treatment for Alzheimer’s with supportive data (discussed further here).
DMSO has unique utility in the disease as DMSO:
•Stabilizes proteins and eliminates misfolded amyloid aggregates (allowing it to remove dangerous amyloid accumulations in a non-injurious way). For example, a computer modeling study indicated DMSO inhibits Aβ aggregation by modulating the stability of the Lys28-Ala42 salt bridge, while molecular dynamics simulations showed DMSO promoted α-helical structure and stabilized Aβ42 (preventing the β-sheet formation that drives toxic aggregation).
•Is an acetylcholine esterase inhibitor,1,2 (the same therapeutic strategy used by the FDA-approved Alzheimer’s drugs donepezil and galantamine).
•Has been shown to increase the activity of lysosomal alkaline phosphatase (ALP) by 20%, which likely enhances the clearance of the toxic protein aggregates that contribute to neurodegeneration.
Note: a nasal insulin DMSO spray developed for treating AD was found in rats to deliver insulin throughout the brain within an hour with no signs of toxicity.1,2,3
Additionally, DMSO’s foundational properties (e.g., improving circulation, reducing inflammation and reawakening dormant “shocked” cells) directly counteract the pathological processes that create AD. For example, DMSO has been shown to inhibit activation of NLRP3 inflammasomes and caspase-1, which mediate the chronic neuroinflammation that accelerates AD progression.1,2,3
Note: DMSO is routinely used in laboratory settings to solubilize and dissolve amyloid peptides for experimental use,1,2,3,4 and initial solubilization conditions (including DMSO exposure) significantly impact the reproducibility of Aβ aggregation kinetics. Studies characterizing the toxicity of different Aβ species (oligomers, protofibrils, fibrils) and molecular weight fractions have relied on DMSO for peptide preparation.1,2 DMSO has also been used as a solvent platform for screening anti-aging drug cocktails against Aβ and tau neurotoxicity in neuronal cell lines.
In animal studies, DMSO has repeatedly demonstrated direct cognitive benefits in AD models:
In rats where intracerebroventricular streptozotocin (STZ) infusions were used to model sporadic AD, daily IV DMSO for 2 weeks counteracted memory impairment, improving performance in behavioral and memory tests. This was corroborated by a study where chronic intracerebroventricular DMSO at 10% (but not 2.5% or 5%) significantly attenuated STZ-induced spatial memory deficits in the Morris water maze, with the authors attributing the benefit to DMSO’s antioxidant, anti-inflammatory, and cerebral perfusion-enhancing properties.1,2 In another STZ study, a separate group found similar trends toward decreased neuroinflammation (IL-1β, TNFα), behavioral improvements and elevated BDNF with DMSO treatment.
In AD model rats with Aβ25-35 injected into the hippocampus, DMSO improved learning and memory, reducing escape latency and search distance in the Morris water maze. Notably, no significant synergistic effect was seen when DMSO was combined with Ginkgo biloba extract,⬖ suggesting DMSO alone was already exerting a substantial effect. In a similar study, DMSO and Ginkgo biloba extract⬖ each improved learning and memory in Aβ25-35 AD model rats, with effects associated with modulation of hippocampal APP expression.
Furthermore, in mice genetically engineered to have AD, DMSO increased spine density in a region-specific manner in the hippocampus, enhanced spatial memory accuracy, modulated olfactory habituation, and displayed an anti-anxiety effect. Despite these improvements occurring in animals with elevated Aβ levels, DMSO did not reduce oligomeric Aβ species, and instead appeared to act through modulation of NMDA receptor signaling (as the NMDA antagonist MK-801 recapitulated DMSO’s effect on spine density) leading the authors to conclude: “DMSO should be considered as a true bioactive compound, which has the potential to be a beneficial adjuvant to counteract Aβ-mediated synaptotoxicity and behavioral impairment.”
In a separate mouse AD study, DMSO independently crossed the blood-brain barrier, improved cognition, and reduced neuroinflammation — effects observed even though the study was designed to test serpina3n, not DMSO itself.
In young mice (3–4 months) genetically engineered to have early-onset AD, 0.01% DMSO mixed with drinking water mitigated the visual declines and retinal thickening seen in the prodromal phase of AD (with benefits comparable to R-carvedilol), indicating DMSO enhanced the energy-dependent transport of water out of the eyes.
In C. elegans, DMSO greatly delayed (by 48–98%) the paralysis caused by Aβ42 and extended lifespan by 23.0–24.4%.1,2 The researchers attributed the delay in paralysis to DMSO modulating neurotransmission, noting DMSO reduced paralysis from acetylcholine-related agents by ~70% and that the protection was dependent on the daf-16/FOXO longevity pathway.
In cell culture, low concentrations of DMSO (0.015625–0.0625%) increased the viability of brain cells and masked Aβ toxicity (concentrations readily achievable in brain tissue from taking DMSO at home or DMSO’s routine use in cellular experiments).
Note: since DMSO is used so frequently in Alzheimer’s research, it has been repeatedly proposed this protective effect likely has contributed to overestimating the efficacy of therapies used with it and underestimating the neurotoxicity of amyloid particles.1,2
Additionally, DMSO shifted amyloid precursor protein (APP) splicing in the adult rat hippocampus to favor the neurotrophic isoform APP-695 (increasing its proportion from 89% to 94%) while reciprocally reducing the pathologic KPI-containing isoforms (APP-751/770) that are typically elevated in AD (again underscoring its utility in the disease). In a controlled cortical impact model of TBI in mice, DMSO (used as the vehicle for glibenclamide) demonstrated unexpected independent beneficial effects in female mice, significantly downregulating pathological TAU and TDP43 proteins across multiple brain regions and helping restore cerebral blood flow (effects the study authors highlighted as novel).
A 1980s Japanese review noted DMSO had been explored for dissolving amyloid deposits in Alzheimer’s, with one report documenting partial improvement in motor function.
Human Studies
18 patients with probable Alzheimer’s were treated in Moldova with DMSO and tested regularly for nine months, with great improvements being noted after only three months of treatment, and becoming especially noticeable after six months. Areas of improvement included memory, concentration, and communication alongside a significant decrease of disorientation in time and space.
Likewise according to Jack De La Torre, the leading researcher on DMSO’s neurological applications: “Dimethyl sulfoxide, a powerful free radical scavenger that is known to increase cerebral blood flow clinically and experimentally during various pathological states involving brain, has been shown to improve cognitive function while stabilizing protein enzymes in Alzheimer’s patients treated with this drug for 6 months.”
Note: I could not tell if De La Torre was referring to the Moldovan study or something else.
Combination Studies in AD Models
A large body of research has evaluated therapeutic agents dissolved in DMSO across various AD models.
APP Processing and Secretase Modulation
A variety of agents dissolved in DMSO have demonstrated the ability to shift APP processing away from toxic Aβ production. Cinnamon bark extract⬖ reduced Aβ40 production by 50–60% through its active compounds medioresinol and cryptamygin A, which decreased β-secretase levels. Levistolide A⬖ (from Danggui-Shaoyao-San) reduced extracellular Aβ1-42 in AD cells and was shown to cross the blood-brain barrier, reaching peak brain concentrations within 30 minutes. Apicidin (an HDAC inhibitor) increased ADAM10 (α-secretase) expression through USF1 and ERK signaling, shifting APP cleavage away from the amyloidogenic pathway. Retinoic acid inhibited γ-secretase activity through ERK activation, while sulfonamide-based γ-secretase modulators shifted Aβ40/42 ratios without the Notch-related side effects of complete γ-secretase inhibition. Additionally, multiple BACE1 inhibitors dissolved in DMSO reduced Aβ1-40 and Aβ1-42 production in neuronal cells (but like other amyloid drugs, failed in subsequent human trials). DMSO has also been used to dissolve ursolic acid,⬖ which was identified through high-content screening as a potent inhibitor of Aβ binding to the CD36 receptor, blocking microglial reactive oxygen species production.
Transgenic AD Mice
DMSO combinations have been extensively studied in transgenic AD mice. Dihydromyricetin⬖ improved Morris water maze performance, decreased Aβ accumulation and senile plaques, and restored autophagic flux by increasing Beclin1, the LC3-II/LC3-I ratio, and reducing P62. Numerous HDAC6 inhibitors alleviated cognitive decline, reduced Aβ levels and plaque deposition, decreased hyperphosphorylated tau, upregulated autophagy proteins, and protected cortical neurons from oxidative damage.1,2 A p38 MAPK inhibitor improved cognition by reducing Aβ, tau phosphorylation, BACE1, and presenilin while increasing synaptic and amyloid-degrading proteins. Astragalin improved spatial learning and memory in mice by reducing Aβ plaque deposition and enhancing autophagic flux via the PI3K/Akt-mTOR pathway. Ligustilide⬖ improved learning and memory, promoted Aβ transport and clearance, and reduced neuroinflammation. Chronic blockade of the TRPA1 channel normalized astrocytic activity, prevented neuronal dysfunction, preserved synaptic integrity, and prevented spatial working memory decline. Kenpaullone improved cognitive performance and reduced Aβ plaques, neurodegeneration, and proinflammatory cytokines, while erlotinib improved short-term spatial memory, increased dendritic spine formation, and reduced tau phosphorylation and aggregation.
Among agents targeting Aβ clearance or reduction, novel c-KIT inhibitors enhanced autophagic clearance of Aβ and tau, eriodictyol promoted microglial Aβ clearance,⬖ menthol⬖ inhibited Aβ polymerization and reduced senile plaques, and picropodophyllin reduced Aβ and hippocampal microgliosis, while ellagic acid⬖ reduced both Aβ and caspase-3. GTM-1, rapamycin, and carbamazepine each improved spatial memory and reduced Aβ42 (with GTM-1 activating autophagy via mTOR).
Among agents improving synaptic function and cognition, a JNK inhibitor reduced inflammation, rescued synaptic proteins, and improved cognition; a PDE4 inhibitor improved memory, synaptic plasticity, and hippocampal signaling; CA140 rescued memory and synaptic function via dopamine D1 signaling; puerarin⬖ restored NMDA receptor activity; and isoliquiritigenin⬖ restored excitatory/inhibitory synaptic balance. A SERCA activator improved memory and motor coordination, while clenbuterol increased PSA-NCAM and improved cognition.
Additional agents working through neuroprotection, neurogenesis, or immune modulation include a LXR agonist (which promoted neurogenesis and upregulated apoE), a CB2R agonist (which shifted microglia from M1 to anti-inflammatory M2), idebenone (which improved spatial memory and mitochondrial function), an estrogen receptor beta agonist, and berberine⬖ (which dose-dependently reduced apoptosis and increased Bcl-2 expression in hippocampal neurons).
Sporadic AD Models
Sporadic AD models (using intracerebroventricular STZ) also responded to combination therapies. A triazine derivative improved spatial learning and increased hippocampal pyramidal layer thickness, canagliflozin improved dendritic morphology, the Croton hirtus extract⬖ MECH reversed learning and memory impairments while reducing Aβ and AChE, and rapamycin reduced hyperphosphorylated tau and mTOR signaling. The MIF inhibitor ISO-1 improved hippocampal-dependent contextual memory and reduced cytokine production, with MIF levels found elevated in early-stage AD patients’ cerebrospinal fluid. The GSK-3β inhibitor SB216763 attenuated tunicamycin-induced UPR, synaptic impairments, and memory deficits.
Acute Aβ Injection Models
Acute Aβ injection models have been used to test a wide range of agents. Piper kadsura ohwi extract⬖ significantly ameliorated ethological deficits from both Aβ oligomers and fibrillar Aβ while reducing neuroinflammation via TLR4/NF-κB/TNF-α.1,2 Rapamycin improved cognitive function, reduced hippocampal Aβ deposition, and increased Homer3 expression. Testosterone reversed spatial learning deficits and neuronal loss via BDNF/CaMKII/CREB signaling (an effect completely blocked by the androgen receptor antagonist flutamide). Curcumin⬖ dissolved in DMSO partially improved spatial learning in AD model rats and also reversed Aβ-induced damage to neural stem cells, improving viability and differentiation markers (Nestin, Tuj-1, GFAP).1,2,3 Pioglitazone dose-dependently reversed Aβ1-42-induced activation of MKK4, JNK1, and c-Jun in rat hippocampus, with most p-JNK immunoreactivity co-localizing with microglia.
Additional agents that improved cognition or reduced AD pathology in Aβ-injected models include Angelica keiskei extract⬖ (which dose-dependently improved memory), low-dose genistein⬖ (which upregulated STAT3 and suppressed caspase-3), betulinic acid⬖ (which improved memory, anxiety, and LTP), DL0410 (a dual AChE/BChE inhibitor that improved learning via CREB/BDNF), B. pendula leaf extract⬖ (which reduced oxidative stress and NF-κB signaling), Huanglian Jiedu Decoction⬖ (which reduced microglial activation), a DAPK1 inhibitor (which reduced NLRP3 inflammasome activation and ameliorated memory deficits), and atorvastatin (which prevented synaptotoxicity and neuroinflammation via p38 MAPK inhibition). Biochanin A⬖ attenuated Aβ-induced neuronal death and improved spatial learning via estrogen receptor-dependent p38 MAPK inhibition, while Perilla frutescens⬖ hexane fraction inhibited Aβ aggregation and protected cells from Aβ toxicity as did a purpose-designed beta-sheet breaker peptide.
Additional agents showing neuroprotective effects in Aβ cell models include Dendrobium nobile alkaloids⬖ (which increased cell survival), neuroprotectin D1 (which reduced Aβ42, tau phosphorylation, and apoptosis while enhancing autophagy), curcumin⬖ (which reduced mitochondrial ROS, enhanced autophagy via Rab proteins, and increased axonal transport),1,2,3 icariin⬖ (which inhibited autophagy and reduced Aβ production via p70 S6K), a DPP-4 inhibitor (which activated PI3K/Akt/GSK-3β and reduced tau phosphorylation), sodium ferulate⬖ (which protected hippocampal neurons via Notch pathway modulation), butylphthalide (which protected endothelial cells from Aβ-induced apoptosis via TLR4/COX-2),1,2 ZL006 (which activated Akt/Nrf2/HO-1), MS-275 and JQ1 (which improved memory via CREB and reduced TNF-α), pioglitazone (which reduced Aβ and tau via PPARγ activation), tetrahydroxystilbene glucoside⬖ (which reduced microglial inflammation and APOE/TREM2 expression), berberine⬖ (which promoted microglial M2 polarization via PI3K-AKT signaling, protecting neurons from Aβ-mediated injury), and 5-HT1B receptor inhibition (which restored Aβ1-42-disrupted eNOS and Akt phosphorylation in human endothelial cells, suggesting this receptor mediates Aβ-induced vascular dysfunction relevant to AD).
Okadaic Acid (OA) Models
Okadaic acid models (which induce tau hyperphosphorylation by inhibiting protein phosphatase 2A) also responded to DMSO-delivered therapies. Trillium tschonoskii⬖ decoction improved spatial learning, increased PP2A activity, and reduced tau phosphorylation while preserving hippocampal neurons. Banqiao Codonopsis pilosula decoction⬖ dose-dependently improved cognition, reduced tau hyperphosphorylation, and increased synaptic proteins and dendritic spine numbers.1,2 In zebrafish, Mansonone G⬖ reversed OA-induced cognitive impairments, improving spatial and recognition memory and restoring cholinergic function. Additional agents include ginsenoside Rb1⬖ (which attenuated tau hyperphosphorylation), ginkgolide⬖ combined with insulin (which synergistically improved learning and reduced neurofibrillary tangles), pilose antler polypeptide⬖ (which activated PI3K/AKT and reduced apoptosis), and sarsasapogenin⬖ (which attenuated tau hyperphosphorylation via Akt/GSK-3β).
Tau Pathology
Several findings directly address tau, a key driver of neurodegeneration in AD:
In female mice following traumatic brain injury, DMSO independently reduced pathological TAU and TDP43 proteins across multiple brain regions, an effect comparable to glibenclamide. EGTA (a calcium chelator) decreased tau phosphorylation at Ser396 in AD mice by reducing calpain 2 and CDK5 expression. Resveratrol⬖ reduced cadmium-induced tau hyperphosphorylation via AMPK/PI3K/Akt activation, while resveratrol⬖ also reduced tau acetylation and phosphorylation in aged rats with postoperative cognitive dysfunction by activating SIRT1 and shifting microglia from M1 to M2a phenotype.1,2 Docosahexaenoic acid⬖ administered after TBI reduced phosphorylated tau and APP accumulation. UCH-L1 inhibition blocked tau aggresome formation by reducing K63-linked ubiquitin chains and impairing HDAC6-tau binding.
In C. elegans (AD strains) hemp extract⬖ (containing both cannabinoids and terpenes, dispersed in DMSO) significantly increased lifespan in tau-model nematodes, with CBD⬖ directly hindering protein aggregate formation.
Oxidative Stress
Ferulic acid ethyl ester,⬖ administered intraperitoneally in DMSO, protected gerbil brain synaptosomes from Aβ-induced oxidative damage, reducing reactive oxygen species, protein oxidation, and lipid peroxidation while boosting antioxidant defenses (HO-1, HSP72) and reducing iNOS. Astaxanthin⬖ dose-dependently improved cognitive performance, reduced Aβ42 and malondialdehyde accumulation, inhibited AChE and MAO activities, and increased Nrf2 and miR-124 expression. In mice, DMSO reduced cerebral ROS by 9.8–79% (greatest effect in young mice) and irreversible oxidative protein damage by 30.7–69% (greatest effect in aged mice); these benefits were further enhanced by curcumin.⬖ In rats, SIRT2 inhibition reduced oxidative stress, increased anti-apoptotic bcl-2, and elevated autophagy regulator beclin-1 in cortex and hippocampus, while melatonin⬖ and curcumin⬖ each reduced lipid peroxidation and SIRT2 expression and increased Nrf2 and antioxidant defenses in the cerebral cortex (though without synergistic effect when combined).
MSM⬖ (DMSO’s oxidized metabolite) ameliorated BPA and radiation co-exposure-induced neurodegeneration, reducing AD markers (Aβ42 by 71.6%, AChE by ~50%, tau phosphorylation by 57%), boosting BDNF (132–160%), suppressing the TREM-2/DAP12 pathway, and reducing Aβ plaques by ~75%.
Note: MSM⬖ crosses the blood-brain barrier rapidly (detectable within ~10 minutes of oral intake), reaching brain concentrations of ~2.36 mM after oral supplementation with a half-life of approximately 7.5 days.1,2
AChE Inhibition and Drug Screening
In addition to DMSO itself being an AChE inhibitor, a variety of AChE inhibitors have been tested in DMSO for AD relevance. Novel tacrine-hydroxamic acid derivatives showed potent dual AChE/HDAC inhibition with Aβ inhibition, while thiazole-based inhibitors outperformed rivastigmine and huperzine-A. Among 21 flavonoids screened, galangin showed the strongest brain AChE inhibition (IC50 ~120 μM). Another screening identified a multifunctional probe that (with DMSO) bound Aβ aggregates, inhibited cholinesterase, and improved cognitive and spatial memory.
Natural Compounds and Model Organisms
A wide range of natural compounds dissolved in DMSO have shown neuroprotective effects in AD models. In C. elegans, olive leaf extract⬖ strongly protected neurons from Aβ-induced degeneration, while 3-hydroxytyrosol,⬖ tyrosol,⬖ saffron,⬖ Polygonum multiflorum,⬖ and Ziziphus jujuba⬖ each also provided significant protection.1 Cannabidiol⬖ extended lifespan and slowed age-related neuronal deterioration in C. elegans and enhanced neurite outgrowth and dendritic spine density in mammalian neurons through the same SIRT1 pathway. Quercetin⬖ and rifampicin individually reduced Aβ plaques, with the combination producing the greatest clearance. Quercetin⬖ and kaempferol⬖ (polyphenols isolated from Ginkgo biloba cell cultures) dose-dependently reduced protein carbonylation in C. elegans, a key marker of aging-related protein damage linked to neurodegenerative diseases. Oxoisoaporphine-lipoic acid hybrids improved learning, memory, lifespan, and AChE inhibition in Aβ42 transgenic Drosophila. In zebrafish, purinergic receptor antagonists (caffeine, ZM241385, DPCPX) prevented scopolamine-induced memory impairment.
In cell and animal models, carvacrol⬖ protected neurons from Aβ-induced neurotoxicity and improved memory in AD rats,1,2 while Ashwagandha-derived withanolides⬖ restored axon/dendrite lengths and synaptic markers in Aβ-injured neurons and reversed memory deficits when given orally. Cynarae folium extracts⬖ exhibited neuroprotective effects against Aβ in both cells and rats, while in drug-resistant Aβ flies, anle138b significantly prolonged survival. Ptychopetalum olacoides extract reversed MK-801-induced amnesia, suggesting involvement of the glutamatergic system in its cognitive-enhancing properties.
Additional natural compounds showing anti-Aβ effects include curcumin⬖1,2 (which suppressed microglial HMGB1/RAGE/NF-κB signaling), bisdemethoxycurcumin,⬖ Ginkgo biloba polyphenols⬖ (which protected dPC12 cells from Aβ damage), Perilla frutescens⬖ (which disaggregated preformed Aβ aggregates), icariin⬖ (which reduced iNOS and TNF-α and improved cognition in LPS-induced AD mice), various edible sprout extracts⬖1,2,3 (with buckwheat sprouts⬖ showing the strongest ROS suppression and Aβ reduction), curcumin⬖ (which, through binding amyloid, enabled in vivo detection of retinal amyloid in live transgenic AD mice via confocal ophthalmoscopy, demonstrating the potential for non-invasive AD diagnosis), and the polyphenols nordihydroguaiaretic acid,⬖ baicalein,⬖ and Padina extract⬖ (which protected cells from Aβ42-induced damage). Curcumin⬖ dissolved in DMSO also achieved high serum solubility (at least 3 mM), effectively inhibiting Aβ plaque formation.
Aging-Related AD Models
D-galactose injection creates accelerated aging in rodents that models age-related cognitive decline. Guilingji⬖ (a traditional formula) significantly improved Morris water maze performance in D-galactose-aged rats, restoring antioxidant activity and normalizing neurotransmitters. Ganoderma tsugae extract⬖ improved locomotion, learning, and memory by increasing brain antioxidant enzymes and BDNF while reducing inflammasome activation. Kurarinone⬖ combined with rehabilitation exercise synergistically improved spatial learning and memory, upregulated synaptic plasticity markers (PSD-95, synaptophysin), and reduced Aβ1-42 deposition via the BACE1/Aβ pathway. Resveratrol⬖ significantly increased SOD and decreased oxidative damage in hippocampal neurons of mice with D-galactose-induced aging. Curcumin⬖ reduced APP and tau expression and suppressed HAT activity in AD rats. Artemisinin⬖ also demonstrated neuroprotective effects in an animal model of AD prevention.
Aluminum chloride, which induces Aβ plaque accumulation and tau phosphorylation, was counteracted by a bithiophene derivative that restored antioxidant defenses, prevented neuronal and synaptic loss, and mitigated AD-related pathology.1,2
Vascular Dementia
Vascular dementia (VaD) results from chronic cerebral hypoperfusion that causes progressive neuronal damage and cognitive decline, making DMSO well suited for the condition.
Two p38 MAPK inhibitors dissolved in DMSO repeatedly improved spatial learning and memory in rats with bilateral carotid artery occlusion-induced VaD, reducing hippocampal apoptosis, extending survival, and decreasing p38 MAPK phosphorylation.1,2,3 Curcumin⬖ dissolved in DMSO dose-dependently improved learning and memory in rats with bilateral carotid artery occlusion-induced VaD, reducing hippocampal pathology and promoting brain cholesterol efflux via the LXR/RXR-ABCA1-apoA1 pathway.1,2,3 Paeoniflorin⬖ improved learning and memory in VaD rats by reducing proinflammatory cytokines and inducing microglial polarization from M1 to M2 via cannabinoid receptor type 2. Cathepsin B inhibition reduced mitochondrial abnormalities, neuronal apoptosis, neuroinflammation, and cognitive dysfunction after transient middle cerebral artery occlusion.
Additional agents improving cognition in VaD models include andrographolide⬖ (which reduced hippocampal apoptosis and increased Bcl-2), PGE1 (which improved learning and memory via the BDNF/TrkB pathway), esketamine (which upregulated BDNF, ERK, and Nrf2), tripchlorolide⬖ (which reduced Aβ, NF-κB, and COX-2), IGF-1 (which improved cognition via Akt/mTOR), intranasal recombinant erythropoietin (which improved escape latency and preserved hippocampal neurons), the Shenmayizhi formula⬖ (which upregulated synaptic proteins and VEGF), resveratrol with SIRT1 activation (which improved cognition and enhanced antioxidant capacity in chronic cerebral ischemia), HDAC inhibitor compound 13 (which activated BDNF isoforms and modulated AMPA receptor signaling), rapamycin (which reversed mTOR-mediated autophagy suppression, reducing Aβ accumulation and tau hyperphosphorylation in diabetic rats after orthopedic surgery), and emodin⬖ (which reduced ROS and inhibited neointimal proliferation in carotid artery stenosis). Quercetin⬖ also alleviated hippocampal demyelination in a bilateral carotid artery stenosis model.
Note: molar loss (tooth extraction) in VaD rats worsened cognitive impairment and increased hippocampal apoptosis and iNOS/p38 MAPK/NF-κB expression; a p38 MAPK inhibitor in DMSO reduced these effects, illustrating that dental health impacts cerebrovascular cognitive decline.
DMSO has also been used to differentiate bone marrow mesenchymal stem cells (BMSCs) into neuron-like cells expressing neuronal markers (using a 2% DMSO/BHA medium), which were then transplanted into VaD rat brains, and BMSCs promoted recovery of an AD cell model by upregulating PDGF-BB via the PI3K/AKT pathway.
Reader Reports
My uncle’s wife has dementia and has been unable to speak for over a year. My mom recently visited them and told them about DMSO. He began to give his wife DMSO orally. After two weeks she began to talk again.
I read the article and began giving it to my 93 year old mother in her juice every morning the end of November. She has had some form of dementia for over 15 years. In the last year or so she would get sundowners really bad and could not follow instructions. Since taking the DMSO, she no longer suffers with sundowners. She is more “with it” and can communicate and laugh with us. Her personality is back. She is able to understand when I am asking her to use the bathroom. She is more cognitive and has started coloring in her coloring books again.
I deeply appreciate your posts on DMSO. You helped bring spontaneous interaction back into the life of my father with Alzheimer’s. As well as many other people I have educated about its uses. God bless you.
Meanwhile, your DMSO information seems to have been a game-changer for him [a stroke patient]. We are now about eight weeks into treatment and I have observed: voice is stronger, speech is markedly improved, appetite has increased, dysphagia has changed dramatically---he is now able to eat normal-sized meals. He now feeds himself almost exclusively, using his affected hand. His sense of taste has started to return.
We have been using several of the therapies you have informed us of. I started putting 2/3 teaspoon DMSO with a teaspoon of galactose⬖ in his coffee each morning. I started giving him 2 teaspoons after breakfast in juice and again 2 teaspoons in the afternoon, with good results. Is he cured? NO but he’s functioning a lot better.
Lastly, a reader shared their partner (75) was diagnosed with an inoperable 8cm glioblastoma causing a slow brain bleed, losing the ability to speak, and right-side paralysis, with a prognosis of 3 weeks to live. After copious topical 99% DMSO, motor function began returning within 24 hours. By week 3, he was eating meals at the table. By week 4, he could walk with a walker. A new CT scan at day 55 showed no brain bleed and reduced tumor metrics.
Multiple Sclerosis
Multiple sclerosis (MS) is an autoimmune disease in which the immune system attacks myelin, the insulating sheath around nerves in the brain and spinal cord. As demyelinated nerves progressively lose their ability to transmit signals, patients develop worsening neurological deficits including impaired mobility, vision loss, pain, and cognitive decline. Despite decades of research, existing therapies primarily suppress the immune attack but do not restore lost myelin, leaving patients with accumulating disability.
DMSO has several properties that make it mechanistically well-suited for MS: it crosses the blood-brain barrier, reduces harmful immune activity, decreases inflammation, improves circulation (including to vulnerable white matter tracts), and as discussed in the protein folding section, acts as a chemical chaperone that stabilizes proteins. Additionally, DMSO inhibited the blood clotting triggered by myelin debris in a dose-dependent manner — significant because when myelin is broken apart by the immune system, exposed phospholipids activate clotting pathways that compromise the blood supply nerves depend on, providing a secondary mechanism for MS neurodegeneration that existing anticoagulants do not target. DMSO also prevented bilirubin-induced toxicity in myelinated axons, suggesting DMSO directly protects myelin from the blood breakdown products released during hemorrhage or inflammatory demyelination (likewise a caspase-1 inhibitor—in DMSO—reversed bilirubin’s NLRP3-mediated damage to hippocampal neurons).
This circulatory perspective on MS also aligns with a broader theory that beyond insufficient blood flow to the brain, impaired venous and lymphatic drainage from the brain also contribute to neurodegeneration — both by retaining toxins and by disrupting the glymphatic system (the brain’s waste clearance pathway), leading to accumulation of pathological proteins like β-amyloid and α-synuclein. In 2009, it was proposed that MS was linked to chronic cerebrospinal venous insufficiency (CCSVI) — poor drainage from the head through the jugular veins — and significant clinical improvements were reported after stenting the jugular veins open (a technique also used for other complex neurological conditions before being restricted due to both pharmaceutical lobbying and legitimate safety concerns). More recently, in China, lymphatic-venous anastomosis (LVA) — a microsurgery that creates direct connections between the neck’s deep cervical lymphatic vessels and adjacent veins to bypass obstructed drainage — has already produced dramatic short-term improvements for dementia patients and is now offered at over 30 surgical centers.1,2,3
Note: this article provides an excellent synopsis of the evidence for CCSVI and the neurological conditions it causes (although our treatment approach differs from the authors).1,2
I am inclined to believe this theory is correct, as we saw many neuroimmune patients make dramatic recoveries from jugular stenting when it was still available. Likewise, pioneering clinicians recently found that COVID vaccine injuries frequently resulted from sudden persistent compression of the iliac vein (which they successfully treated with stenting). My own belief is that drainage obstructions often result from poor zeta potential — which both drives fluid flow and creates the expansive force that keeps vessels open — or from inherently weak blood vessels that collapse easily. This explains why hypermobile patients are typically far more sensitive to pharmaceutical injuries (particularly vaccines that impair zeta potential, or via the spike protein, directly weaken blood vessels), and why MS has been found to be 10–11 times as common in hypermobile EDS patients.
Due to the risks associated with stenting, I have pursued other approaches to address impaired drainage (with success). Since DMSO increases lymphatic circulation, improves venous drainage, and clears the pathological protein aggregates that accumulate when drainage fails (as detailed earlier in this article), it may address this underlying impairment through pharmacological rather than surgical means.
Note: pioneering physician Dietrich Klinghardt extensively investigated CCSVI across neurological conditions and reported it present in 100% of tested MS, autism, Parkinson’s, ALS, and Lyme patients — with patients suffering from fibromyalgia and chronic fatigue also frequently presenting with it — and Lyme patients typically testing worse than MS patients (4 of 5 diagnostic parameters positive vs. 2 of 5 in MS).1,2,3,4 He concluded CCSVI was driven by chronic endothelial infections (Borrelia, Rickettsia, Chlamydia pneumoniae, Babesia) that caused inflammation, scarring, and permanent vasoconstriction of the jugular veins1,2,3 (and I would argue zeta potential impairment—as Knisely observed malaria [which is similar to babesia] ultimately killed by life-threatening blood sludging which began with small sludges forming along the vessels within which it hid from the immune system1,2,3). While balloon dilation produced roughly one-third significant improvement, one-third modest, and one-third no lasting benefit, Klinghardt preferred it over stenting because stents caused scarring and vein occlusion1,2 and he found that results were inconsistent regardless of method if the underlying infections, existing tissue compressions and elevated inflammatory markers (TGF-β1, C4a, MMP-9) were not addressed first, as the veins would re-stenose.1,2,3,4,5,6 Likewise, other clinicians at a CCSVI summit corroborated it contributed to neurodegeneration across multiple conditions1,2 and a diagnostic protocol Klinghardt helped create used therapeutic ultrasound to mobilize compartmentalized pathogens from the venous endothelium (or possibly sludges lining the endothelium) into the urine where they could be identified by DNA testing (and often were).1
DMSO in MS Patients
In a famous case from the DMSO literature, Stanley Jacob decided to risk giving a 29-year-old woman paralyzed from MS who also had kidney failure oral DMSO. “Her improvement was dramatic — as dramatic as any benefit I have ever seen,” Jacob reported. Her kidney problems came under control, and after a few more weeks, she walked again. Six years later she still walked, drove her car, and cared for her family — though she was slowly declining (indicating DMSO was not enough to completely treat her condition).
Another more recent case involved a California woman confined to bed in the fetal position at a convalescent hospital, expected to die within months. After starting DMSO through multiple routes (injectable, oral, and topical), she began regaining sensation in her legs — initially experiencing pain, which was considered positive given her prior lack of feeling. Over the following year she regained leg movement and the ability to feed herself, with continued improvement until she was moved to another state where the treatment was unavailable.
The only formal study I know of evaluated 34 MS patients in Russia in 1984. Overall, the investigators felt DMSO had a very positive result, with the best outcomes in patients with remitting MS, while results were more inconsistent in rapidly progressive MS. They attributed the benefits to DMSO causing remyelination, reducing edema, improving nerve cell communication, and having positive effects on immunity and tissue repair, with no side effects reported.
Additionally:
•A Russian case report documented successful DMSO therapy in an MS patient with trophic lesions and a cystostoma.
•DMSO has also been used topically (mixed with anesthetics) on trigger zones to manage trigeminal neuralgia in MS patients, with outcomes including reduction of carbamazepine to minimum doses or complete discontinuation and achievement of full remission during exacerbations.
•A study of 35 MS patients (disease duration 2–18 years) recommended DMSO-containing topical gels mixed with nonsteroidal analgesics for chronic neuropathic pain, myofascial pain from spastic muscles, and pain along peripheral nerve trunks in bedridden patients.
Reader Reports
A few readers have also reported improvements in MS following DMSO use which corroborate the previous data.
One reader’s wife with MS-related trigeminal neuralgia that had been painful for over a year and a half tested 70% DMSO cream on a small spot on her face, and her pain dropped 90%. The next morning she applied it over the entire trigeminal area and the pain was 99.9% gone — even after three days without reapplication, the pain had not returned.
Another reader reported that DMSO “brings my MS hugs to a standstill. Before my husband found this for me I just had to endure them or go to the hospital for morphine.”
A reader with MS, fibromyalgia, liver fibrosis, CRPS, and lymphedema described oral and topical DMSO as “a godsend” after about a year of use.
An MS patient taking oral DMSO four times daily reported it was “incredibly helpful for pain and energy.”
One reader reported that MSM⬖ (DMSO’s oxidized metabolite), along with other supplements, allowed them to stop all prescribed MS medications and their associated side-effect medications, stating they “feel younger and better than ever.”
Note: a 1982 article stated that DMSO had been touted as a miracle drug for numerous conditions including multiple sclerosis—suggesting these readers’ experiences are not unique.
DMSO Myelin Interactions
•X-ray diffraction studies have revealed that DMSO directly interacts with myelin structure and at high concentrations DMSO produces a fully reversible transformation of nerve myelin into a new highly ordered structure (DMSO squeezes the water out between myelin wraps and selectively alters the outer membrane layer).1,2
•Clemastine, an antihistamine being investigated as a remyelinating agent for hypomyelinating diseases, also showed preliminary effects on myelination processes in vitro.
•Oligodendrocytes are the cells that produce myelin in the central nervous system, and their regeneration is essential for remyelination. DMSO, in turn, has been shown to drastically increase the generation of oligodendrocyte precursor cells from human stem cells1,2,3 (and for this effect to be further enhanced in combination with all-trans-retinoic acid).1,2 Additionally, in neural stem cells, γ-secretase inhibitors (in DMSO) modulated Notch1 signaling and increased oligodendrocyte marker expression and likewise, clobetasol (in DMSO) promoted neural stem cell differentiation into both neurons and oligodendrocytes.
Note: one study found DMSO shifted stem cells towards producing astrocytes, suggesting lower doses promote oligodendrocyte differentiation, while longer sustained ones promote astrocytes.
Other Demyelinating Diseases
Experimental autoimmune encephalomyelitis (EAE), the standard animal model for MS, immunizes animals against myelin proteins to trigger autoimmune demyelination, and hence provides poignant insights for treating MS. Notably, as I show here, the hepatitis B vaccine (which overlaps with myelin, creating autoimmunity to myelin) has been repeatedly linked to multiple sclerosis and also has caused autoimmune EAE.
In a 1969 study DMSO was listed as a therapeutic option for EAE, and in EAE experiments, animals are frequently treated with a therapeutic agent dissolved in DMSO. Across these studies, a consistent pattern emerged: agents dissolved in DMSO improved EAE clinical scores, decreased inflammatory cell infiltration, and reduced demyelination — with individual agents distinguished by their specific mechanisms and additional effects.
Nordihydroguaiaretic acid,⬖ tanshinone IIA,⬖ and sulforaphane⬖ increased protective antioxidant and anti-inflammatory signals while lowering inflammatory IL-17A in EAE immune cells.⬖ Nordihydroguaiaretic acid⬖ also restored the blood-brain barrier, and in a separate study, both nordihydroguaiaretic acid⬖ and tanshinone IIA⬖ promoted remyelination and recovered axon integrity.1,2,3,4,5 Curcumin⬖ delayed disease onset and in addition to reducing inflammatory infiltration and myelin loss, normalized impaired autophagy and lowered inflammatory cytokines.1,2 Mitochondrial division inhibitor 1 decreased oligodendrocyte and spinal cord apoptosis, protected axonal integrity, and promoted nerve growth factors.1,2 Butylphthalide protected mitochondria, inhibited necroptosis, and lowered TNF-α and IL-1β, while ferrostatin-1 (a ferroptosis inhibitor) and matrine⬖ each similarly reduced neurological severity.1,2 An epoxide hydrolase inhibitor reduced disease severity in chronic EAE by suppressing multiple inflammatory pathways (TLR4/NF-κB signaling, inflammasomes, COX-2, and nitric oxide synthases) and in a separate study, promoted remyelination via enhanced myelin proteolipid protein expression.1,2
Additional agents demonstrating efficacy in EAE include hydroxyfasudil (which also prevented EAE onset and suppressed key pro-inflammatory cytokines), a p38 MAPK inhibitor1,2 (which reduced IL-17 expression), ginkgolide A⬖ (which delayed disease onset and shifted macrophages toward an anti-inflammatory state), isoliquiritigenin⬖ (which suppressed inflammatory Th1 cells and activated protective astrocytes), nitidine chloride⬖1 (which increased anti-inflammatory IL-10 when given preventively), a JAK2 inhibitor (which delayed onset and decreased Th17-driven inflammation) and arctigenin⬖ (which decreased Th1 and Th17 cells via AMPK/PPAR-γ activation and p38 inhibition), a caspase-1 inhibitor (which delayed onset and lowered IL-1β and IL-18), and schisandrin B,⬖ baicalin,⬖ AG490, and NBP.1,2,3
In addition to EAE models, in cells from newly diagnosed and IFN-β-treated MS patients, silymarin⬖ suppressed Th1 cell proliferation and inhibited IFN-γ production in a dose- and time-dependent manner, suggesting it could complement existing MS therapies. Chrysin⬖ reduced disease severity and demyelination in experimental autoimmune neuritis (an animal model of Guillain-Barré syndrome).
Lastly, one reader with Morvan’s syndrome, a rare and severe autoimmune channelopathy (that developed following viral encephalitis) reported that after two weeks of topical 70% DMSO cream, inflammation in his hands visibly decreased and pain was reduced by 75%. His case was particularly severe, having required multiple nerve decompression surgeries, four joint replacements, cervical fusion, and two nerve stimulator implants.
Cuprizone Models
Cuprizone is a copper chelator that, when fed to mice, causes toxic demyelination — providing a model to study remyelination independently of the autoimmune component. In cuprizone-demyelinated mice, the cannabinoid agonist WIN55212-2 (1 mg/kg in DMSO) significantly reduced weight loss, improved motor function, reduced corpus callosum demyelination, increased myelin basic protein expression, decreased astrocyte activation, and upregulated juxtanodin and Nkx2.2 (transcription factors involved in myelin repair).1,2,3 Hydroxyfasudil and fasudil also improved behavioral deficits, attenuated demyelination, and strongly inhibited microglia-driven neuroinflammation in the cuprizone EAE model. Mitochondrial division inhibitor 1 protected oligodendrocytes and suppressed complement activation on oligodendrocytes in the corpus callosum. Biochanin A⬖ improved grip strength, spatial and recognition memory, and mitigated hippocampal and prefrontal cortex neuronal damage.
Neuromyelitis Optica
Neuromyelitis optica (NMO) is a related autoimmune demyelinating disease that preferentially attacks the optic nerves and spinal cord through antibodies targeting aquaporin-4 water channels. In an NMO mouse model, tanshinone IIa⬖ (in DMSO) significantly reduced loss of aquaporin-4, GFAP, and myelin basic protein, decreased neutrophil and microglial infiltration, and increased neutrophil apoptosis. Arbidol (in DMSO) similarly increased cortical cell survival from 49% to 79% in vitro and reduced aquaporin-4/GFAP loss in vivo.
White Matter Injury and Neonatal Myelination
Several studies have demonstrated that agents combined with DMSO can protect or restore myelination in non-autoimmune white matter injury. The hydrogen sulfide donor anethole trithione promoted remyelination, oligodendrocyte precursor cell proliferation and differentiation, microglial myelin debris clearance, and long-term functional recovery after subcortical white matter stroke. Siponimod reduced myelin loss and brain atrophy after intracerebral hemorrhage. An α5 GABAA receptor inverse agonist promoted oligodendrogenesis, myelination, and improved motor function in ischemic rats. In a neonatal white matter damage model, miconazole (in DMSO) significantly increased myelin basic protein expression and improved myelin ultrastructure. MiR-219 agomir improved oligodendrocyte maturation in neonatal rats with LPS-induced neuroinflammation via the ERK1/2 pathway. In spinal cord injury, an epidermal growth factor receptor inhibitor reduced demyelination and myelin-associated inhibitory molecule expression.
DMSO and Protein Folding
Chemical chaperones are small molecules that help proteins be folded into their correct configuration, and hence can ensure protein stability or help the body eliminate misfolded protein. Since many challenging diseases (particularly genetic ones) are a result of misfolded or non-functional proteins, chemical chaperones offer a potentially invaluable therapeutic strategy.
Note: I believe the physiologic zeta potential has an important role in ensuring the correct folding of proteins.
Some of the best-known chemical chaperones include glycerol, deuterated water, and DMSO (which is thought to be in part due to it creating a tighter packing around proteins and stabilizing their conformation). DMSO, in turn, has shown promise in misfolding diseases such as nephrogenic diabetes insipidus,1,2 cystic fibrosis and Machado-Joseph disease, and can increase the ability of impaired immune cells (due to defective HLA-DM) to present the antigens necessary to mount an immune response.1
One protein-misfolding disease, amyloidosis, occurs when misfolded proteins enter the bloodstream, aggregate into amyloid fibrils, and deposit in tissues (which over time damages organs and can lead to organ failure). As options for this disease are limited, DMSO was attempted, and early positive responses led to it being repeatedly used in certain areas (e.g., in Japan), where it was found the best results were obtained when high concentrations could be applied directly to the affected area (e.g., bladder lining or skin), and that while not consistently effective, a large body of literature showed it repeatedly produced dramatic results for patients with otherwise fatal conditions, and appeared to do so by both dissolving amyloids and facilitating their urinary excretion (detailed here).
Note: since many cancer-causing proteins are misfolded proteins, it is thought that this may partly explain DMSO’s anticancer properties.
In addition to diseases discussed previously, three neurological conditions characterized by defective proteins have shown promising responses to DMSO.
Niemann-Pick disease
Niemann-Pick disease is a rare genetic lysosomal storage disorder marked by intracellular accumulation of sphingomyelin, either due to a defect in sphingomyelinase, the enzyme which breaks it down (types A/B) or defective lysosomal cholesterol trafficking (type C). Each causes damage to the organs and neurodegeneration, with A [NPA] being the most severe (death is typical at 2-3 years), B [NPB] the least severe (minimal neurological involvement), and C [NPC] on average causing death around 13 years of age. As DMSO stabilizes proteins and improves cellular transport, it has been repeatedly explored for this condition (particularly type C).
This disease is frequently studied in human fibroblasts with that defect. There DMSO has been shown to do the following:
•In normal fibroblasts, 2% DMSO for 4 days increased sphingomyelinase activity to 230% of control levels in normal cells in a dose-dependent manner (with a much smaller increase in other lysosomal hydrolases). In fibroblasts from NPC patients, 2% DMSO raised sphingomyelinase activity by 480% (correcting the deficiency), along with a smaller (280% increase) seen in NPB fibroblasts.
Note: in NPC, a decrease in sphingomyelinase activity results from the massive buildup of cholesterol in the cell (rather than genetic impairment of the enyzme).
•In normal fibroblasts, 2% DMSO increased sphingomyelinase activity, primarily within the lysosomes, by 168-263% (depending on pH). In NPC fibroblasts (which retained 30% sphingomyelinase activity), DMSO caused a 2+ fold increase in activity that brought it close to normal levels, whereas in NPA and NPB fibroblasts no improvement was seen. Additionally, smaller increases in other lysosomal hydrolases were seen, indicating DMSO specifically targets sphingomyelinase.
•When NPC fibroblasts were exposed to LDL, 2% DMSO had minimal effect in the first 12 hours but substantially reduced the excessive accumulation of unesterified cholesterol in lysosomes thereafter (particularly at 24–48 hours), bringing cholesterol levels close to those in normal cells (markedly diminished perinuclear/lysosomal cholesterol fluorescence), and restored the impaired cellular homeostatic responses to cholesterol.
•In normal fibroblasts, twelve specific drugs (e.g., older psychiatric ones) were found to markedly reduce cholesterol esterification (e.g., one caused a 93% reduction for at least 48 hours). DMSO (2%) partially counteracted this and restored 16-20% of the lost cholesterol processing capacity, suggesting potential use in NPC.
•2% DMSO (but not 1% or 4%) significantly increased sphingomyelinase activity in both normal and NPC fibroblasts. However unlike the other studies, none of the concentrations reduced cytoplasmic cholesterol in NPC fibroblasts.
•Lastly, in mice fibroblasts (modeling NPA/B), 2% DMSO caused a 2-3 fold increase in sphingomyelinase activity.
These results suggested DMSO could partially improve the disease, and they (especially the early ones) generated sufficient interest to explore using DMSO to treat it where the following was found:
In NPA/B mice, oral DMSO administration (0.25% in drinking water from 6 weeks of age) prolonged survival from ~12 weeks in untreated mice to an average of 15 weeks and 5 days (maximum 17 weeks and 5 days) and delayed the onset of neurological symptoms such as tremors. However, it failed to prevent symptom progression or improve manifestations once present and had minimal effect on cholesterol accumulation (liver slightly decreased, spleen and brain were unaffected).
•In NPC mice, flavopiridol (dissolved in DMSO) and infused directly into the brain dose dependently reduced hyperphosphorylation of cytoskeletal proteins and improved behavior.
•In human NPC patients, a Japanese paper reported oral administration of DMSO resulted in some clinical benefits, including decreased size of hepatosplenomegaly, lesser frequency of seizures, and improved EEG findings. However, the progressive clinical course of the disease was not changed, although it appeared to slow down.
The author of that paper also published:
•A case report of an 8 year old girl with NPC who presented with severe psychomotor deterioration, marked cortical atrophy, frequent seizures, moderate ventricular dilatation and hepatosplenomegaly. Oral DMSO was shown to normalize her sphingomyelinase activity. After 2 years of oral DMSO, she had a slight improvement in response to her surroundings, her seizures decreased (allowing an anticonvulsant to be tapered), her EEGs showed marked improvement (theta waves and spindles normalized), there was no further progression of her cortical atrophy, and both the spleen and liver shrank to normal size.
•Another report on siblings with NPC who clinically improved with DMSO.
•A study that again found DMSO corrected sphingomyelinase deficiency in cultured fibroblasts from NPC patients.
•A study showing other dipolar aprotic substances besides DMSO also selectively increased sphingomyelinase activity.
Note: lastly, a final case report detailed unsuccessful treatment of NPC with oral DMSO.
Creutzfeldt-Jakob Disease and Prion Disorders
Prion diseases (such as CJD in humans and scrapie in animals) result from normal prion proteins misfolding into a pathogenic form (PrPSc) that aggregates into toxic fibrils and progressively destroys brain tissue. These conditions are universally fatal and have no approved treatment.
DMSO has shown therapeutic potential through multiple mechanisms. As a chemical chaperone, DMSO stabilized the normal alpha-helical structure of newly synthesized prion protein in infected neuroblastoma cells, inhibiting its conversion to the pathogenic form. In cells carrying a familial CJD mutation, DMSO reversed the mutant phenotype by reducing lysosomal accumulation of misfolded prion protein and restoring its normal distribution to the cell surface.1,2 In yeast prion models, DMSO cured multiple prion variants more efficiently than guanidine hydrochloride by enhancing chaperone activity (Hsp104) and converting aggregated protein back into soluble forms.
DMSO also partially inhibited PrPSc aggregation, producing amorphous aggregates with less than 1% of the prion infectivity of untreated rod-like aggregates when inoculated into hamsters.1,2,3 In prion-infected hamsters, DMSO prolonged incubation time, reduced the rate of PrPSc accumulation in the brain, and (like seen in amyloidosis) increased urinary clearance of protease-resistant prion protein — though it was not sufficient as a standalone cure.1,2
Ruthenium-DMSO complexes (related to NAMI-A) showed even stronger effects, disaggregating prion peptide fibrils, reducing copper-mediated oxidative stress, and rescuing neuronal cell viability from 33% to 85%.1,2
One reader reported that her cousin with CJD improved after approximately one month of IV DMSO infusions; we subsequently learned she operated an infusion center and had used high-dose vitamin and DMSO infusions to treat numerous severe conditions, with her cousin still alive several years later.
Note: two prion studies found less favorable results,1,2 indicating results may vary by species, strain, or protocol.
Lastly, DMSO’s protein-stabilizing properties likely extend beyond misfolded proteins — it has been shown to act as a pharmacological chaperone that restores normal membrane receptor trafficking, and (in many studies) to stabilize brain microtubules during isolation by preventing tubulin depolymerization under stress.1,2 Since microtubules are essential for neuronal function and are impaired in neurodegenerative diseases, this likely enhances DMSO’s neuroprotective effects in conditions like strokes or TBIs, where microtubule collapse from oxidative stress or ischemia worsens neuronal damage.
Note: the benefits seen in the final disease, Down Syndrome, (discussed further below) may result from DMSO’s neuroprotective and antioxidant effects, combined with DMSO’s ability to act as a chemical chaperone (which could help cells manage proteostasis overload from excess proteins and support clearance of misfolded or damaged proteins), or DMSO improving transport of vital nutrients across compromised membranes.
Cognitive Impairment and Dementia
The same degenerative processes which cause dementia initially trigger cognitive impairment and as such, being able to head this off early, beyond greatly improving one’s current quality of life, also allows much worse situations to be avoided. As these neurodegenerative processes (e.g., poor blood flow and inflammation) also underlie many of the other neurological disorders DMSO treats, significant data has accumulated for either DMSO alone, or DMSO in combination with another therapy to reverse cognitive loss.
The animal research in this field is as follows:
When rats had their carotid arteries surgically modified to significantly reduce the amount of blood going to their brain, after 3 months, it was found that DMSO prevented both the neuronal damage and the significant loss of spatial memory and learning that otherwise resulted from that chronic loss of cerebral blood flow.
In a similar study, rats 14 weeks old were subjected to either permanent bilateral carotid artery occlusion or sham occlusion (mimicking the chronic vascular impairments many experience with increasing age) and then tested for visuospatial memory function. After 14 weeks, four rats who had shown persistent and severe memory impairment received DMSO and FDP for 7 days, which improved their memory by 54%, almost reaching the cognitive function of the controls. Unfortunately, this improvement was partially lost once DMSO-FDP were discontinued (as the blood supply was still occluded).1,2
Lurcher mice are used to study olivary and cerebellar disorders because their Purkinje cells can’t survive (e.g., by 30 days of age their walking is grossly abnormal). When these mice received DMSO, it prevented the age-related deterioration of certain cognitive functions (e.g., memory and spatial learning abilities).1,2
DMSO has also demonstrated direct cognitive effects in other contexts. It reduced MK-801-induced neuronal necrosis in the rat cingulate and retrosplenial cortex by 80–86%, even when administered up to 4 hours after dosing—a finding relevant to both anesthesia safety and neuroprotection. In morphine-sensitized mice, DMSO independently enhanced spatial memory formation in the Morris water maze (with the effects further enhanced by curcumin⬖). In a mouse model of thiamine⬖ deficiency (relevant to Wernicke’s encephalopathy), DMSO partially improved behavioral deficits and reduced thalamic damage, and when combined with high-dose thiamine,⬖ enhanced recovery beyond thiamine⬖ alone. DMSO alone also produced no spatial learning deficits when injected into the hippocampus in an 8-arm radial maze task, confirming its safety as a vehicle in cognitive research.
Anesthesia-Induced Cognitive Impairment
General anesthesia, particularly prolonged or repeated exposure, can cause lasting cognitive deficits and is recognized to significantly increase the risk for Alzheimer’s (making it a growing concern for both young children whose brains are still developing and the elderly). As we’ve seen numerous people tip over into dementia (as some of their neurons which are put to sleep during the surgery never seem to wake up afterwards), we hence try to minimize surgeries, have anesthesiologists use less toxic anesthetics (inhaled anesthetics cause the most issues), and use nutraceuticals to counteract that sedation.
Fortunately, DMSO addresses both the recognized mechanisms of anesthesia toxicity (e.g., neuroinflammation, oxidative stress, mitochondrial dysfunction, or neuronal apoptosis) and what we believe the key issue is (cells becoming stuck in a dormant phase). Unfortunately, all existing research on this subject involved DMSO combined with another agent, so definitive proof DMSO alone antidotes anesthesia toxicity does not exist.
Sevoflurane, one of the most widely used inhalational anesthetics, has been the most extensively studied. Resveratrol⬖ dissolved in DMSO repeatedly protected against sevoflurane-induced cognitive deficits in both aged and neonatal rats by activating the SIRT1 pathway, reducing neuroapoptosis, upregulating BDNF, and improving spatial memory.1,2,3 Additional combinations protecting against sevoflurane-induced cognitive impairment include arctigenin (which reduced inflammation via the Akt/NF-κB pathway and upregulated CTRP6), honokiol⬖ (which activated SIRT3, reducing neuroinflammation, oxidative stress, and mitochondrial dysfunction), SAHA (which inhibited NLRP3 inflammasome activation and ameliorated cognitive impairment in aged mice),1,2 curcumin⬖1,2 (which improved memory retrieval dysfunction and reversed sevoflurane-induced Aβ increases and BACE-1 upregulation in aged rats), a S1P agonist (which prevented hippocampal apoptosis in neonatal rats), progesterone (which attenuated sevoflurane-induced neuronal injury in primary hippocampal neurons via progesterone receptors and Akt signaling), and inhibitors of the TLR4-p38MAPK-NF-κB pathway (which attenuated cognitive decline).
Note: in one neonatal rat study, resveratrol⬖ improved molecular markers (SIRT1, PGC-1α, FOXO3α, SOD) and reduced brain pathological damage after sevoflurane, but Morris water maze testing showed no significant behavioral improvement across groups.
Propofol, another widely used anesthetic, causes dose-dependent neuronal apoptosis and lasting cognitive deficits with repeated neonatal exposure. Agents dissolved in DMSO that protected against propofol neurotoxicity include dexmedetomidine (which activated PI3K/Akt/GSK-3β in multiple studies),1,2 etanercept (which blocked TNF-α-mediated damage), SAHA (which reversed learning and memory impairments by modulating histone acetylation, synaptic proteins, and CREB phosphorylation), 17β estradiol (which reversed deficits through the ERK pathway), hydroxylfasudil (which reduced apoptosis via p38 MAPK inhibition and improved Morris water maze performance) and coenzyme Q10⬖ (which partially reversed propofol-induced mitochondrial respiratory chain inhibition in primary hippocampal neurons).
Isoflurane-induced cognitive impairment was counteracted by dexmedetomidine dissolved in DMSO (which activated PI3K/Akt and reduced hippocampal apoptosis), intranasal levosimendan (which protected newborn rat brains), and modafinil (which improved novel object recognition and contextual fear memory during the awakening period). Dexmedetomidine in DMSO also dose-dependently protected neonatal rat hippocampal neurons from etomidate-induced injury via the same PI3K/Akt pathway.
Other anesthetics: Dexmedetomidine attenuated ropivacaine-induced mitochondrial toxicity via p38 MAPK. Curcumin⬖ in DMSO also protected developing rat brains against ketamine-induced neurotoxicity through the Nrf2-ARE antioxidant pathway, and also protected neonatal hippocampal neurons against the neurotoxicity of the anticonvulsants phenobarbital and valproic acid, significantly reducing neuronal loss.1,2
Postoperative cognitive dysfunction (POCD), which affects a significant percentage of elderly surgical patients, similarly responded to DMSO-delivered agents. Jasplakinolide (an F-actin stabilizer) reversed POCD deficits in aged mice by restoring dendritic spine numbers, glutamate receptor expression, and hippocampal neuronal activation. Other agents improving POCD include a P300 inhibitor (which improved water maze performance and reduced hippocampal apoptosis after cardiopulmonary bypass), ononin (which reduced neuroinflammation and oxidative stress after nephrectomy in aged mice), and the Hippo/YAP pathway modulator Iduna. In a series of experiments, rapamycin activated hippocampal autophagy to alleviate postoperative cognitive impairment in aged rats, exendin-4 protected cognition by stabilizing blood glucose, and propofol at therapeutic doses prevented excessive autophagy from electroconvulsive shock (each through complementary mechanisms).
Other Causes of Cognitive Impairment
A wide range of conditions impair cognition, and in many of these, therapeutic agents delivered in DMSO have demonstrated cognitive protection.
Sleep disorders are a significant and underappreciated cause of cognitive decline. In sleep-deprived rats, diallyl sulfide⬖ dissolved in DMSO reversed cognitive impairment, reducing anxiety behaviors and hippocampal oxidative stress, while resveratrol⬖ prevented sleep deprivation-induced cognitive deficits by reducing neuroinflammation and enhancing synaptic plasticity proteins. In chronic intermittent hypoxia models (which simulate obstructive sleep apnea), agents improving cognition include a TGF-β1 inhibitor (which improved spatial memory by reducing hippocampal TGF-β1/Smad3 signaling), capsaicin,⬖ STAT3 pathway inhibitors, an α7 nicotinic acetylcholine receptor agonist (which upregulated BDNF and CREB), and adenosine A1 receptor antagonists.1,2
Diabetic cognitive impairment was improved by a variety of agents in combination with DMSO. Rapamycin improved cognition across multiple studies by reducing mTOR, hyperphosphorylated tau, and apoptosis,1,2,3 while captopril and losartan improved cognitive function through a distinct mechanism — inhibiting the ACE-angiotensin II-AT1R axis in the hippocampus of diabetic mice. A TRPV4 agonist reversed cognitive deficits and enhanced hippocampal neurogenesis, and CNTF improved escape latency and increased hippocampal choline acetyltransferase expression via JAK2/STAT3. Additional agents improving diabetic cognitive impairment include pentamethylquercetin⬖ (Akt/CREB signaling and increased synaptic proteins), sulforaphane⬖ (PI3K/Akt/GSK-3β and inhibited mitochondrial apoptosis), curcumin nicotinate⬖ (improved spatial learning by regulating autophagy), krill oil/EPA⬖ (NRF2 antioxidant pathway), Urtica dioica⬖ and pioglitazone (oxidative stress markers and mitochondrial function), and PSD-95/nNOS inhibitors (reduced neuronal injury via Sirt3).
Sepsis-associated encephalopathy responded to a HIF-1α inhibitor (which improved cognitive function and reduced autophagy), resveratrol⬖ (which activated SIRT1, reduced hippocampal apoptosis and microglial activation), ROCK inhibitors (which reduced cerebral injury, inflammation, and improved spatial memory), and molecular hydrogen (which improved survival and cognition via Nrf2-dependent signaling). Additional agents improving cognition in other disease states include Babao Dan⬖ for minimal hepatic encephalopathy, hesperidin⬖ for hyperhomocysteinemia and curcumin⬖ for cerebral hypoperfusion combined with diabetes.
Epilepsy-related cognitive impairment was improved by rolipram (a PDE4 inhibitor that rescued spatial learning, hippocampal long-term potentiation, and phosphorylated CREB after status epilepticus),1,2,3 an eEF2K inhibitor (which restored AMPKα1 expression and improved cognition and social behavior in chronic epileptic mice), semaglutide (which improved cognitive function and protected hippocampal neurons via SIRT-1/NLRP3), and perampanel (which improved spatial learning and reduced hippocampal damage in immature mice with temporal lobe epilepsy).
Neuropathic pain frequently impairs cognition, and this was reversed by an α5-GABAAR inverse agonist (which restored GABAergic signaling and improved recognition and spatial memory in sciatic nerve injury rats), SAHA (which improved spatial learning in bone cancer pain rats), and curcumin⬖ (which enhanced hippocampal neurogenesis and synaptic plasticity). Rutin⬖ also protected against cadmium-induced cognitive impairment by restoring antioxidant balance.
Neonatal brain injury from hypoxia-ischemia was treated with coumestrol⬖ (which partially reversed cognitive deficits, tissue loss, and reactive astrogliosis), C2-ceramide (which reduced neonatal hypoxic-ischemic brain damage by 45–65%) and recombinant human chemerin (which improved both short-term neurological function and long-term cognitive outcomes via the ChemR23/CAMKK2/AMPK/Nrf2 pathway). Repetitive transcranial magnetic stimulation combined with BDNF/TrkB signaling similarly improved spatial learning in rats with prenatal stress-induced cognitive deficits.
Oxidative stress and neuroprotection were addressed by ferrostatin-1 and a caspase inhibitor (which improved cognitive function and preserved synaptic proteins in iron-overloaded rats), and by glutamate receptor antagonists (which protected white matter and myelin ultrastructure in a focal ischemia model relevant to vascular cognitive decline).
Agents addressing psychiatric-related cognitive impairment include a CB1R antagonist (which rescued electroconvulsive shock-induced memory deficits without affecting antidepressant efficacy), fisetin⬖ (which reversed MK-801-induced cognitive impairment in a schizophrenia model by restoring ERK/CREB/CaMKII), and 7,8-dihydroxyflavone⬖ (which improved spatial cognition and restored hippocampal dendritic spines in a schizophrenia model).
Additional agents improving cognition across various models include a complement C3a receptor antagonist (LPS-induced cognitive dysfunction), a GPER agonist (traumatic brain injury), novel PDE4 inhibitors roflupram and chlorbipram (which reversed scopolamine-induced cognitive deficits without the emetic side effects of existing PDE4 inhibitors), and pregnenolone⬖ (which improved spatial learning and upregulated choline acetyltransferase in aged rats). Sonchus asper⬖ and Launaea⬖ procumbens extracts each improved cognition and brain antioxidant status (with Sonchus asper⬖ also inhibiting acetylcholinesterase), while 7,8-dihydroxyflavone⬖ enhanced response learning via TrkB receptor phosphorylation and epigallocatechin gallate⬖ reversed high-fat diet and rapamycin-induced cognitive impairment while improving serotonin and acetylcholinesterase activity.
Human Studies
These findings have been replicated in humans. A study evaluated 104 elderly adults with organic brain disease due to cerebrovascular diseases (e.g., a previous stroke, cerebral embolism, or hardening of the arteries of the brain), a previous head injury, senility, or degenerative disease (e.g., Parkinson’s, hyperthyroidism, or epilepsy). They received two DMSO mixes, Merinex (DMSO with amino acids⬖) and Ipran (DMSO with vasoactive substances), typically alternating between the two, and for the majority of the time as an injection, and a minority of the time orally (with the fastest results occurring if both routes were used simultaneously), all of which resulted in remarkable improvements.1,2 To quote the author:
The DMSO aminoacid therapy is undoubtedly valuable in the treatment of numerous organic cerebral diseases. At the same time, thanks to the improved cerebral blood irrigation achieved by DMSO used in combination with vasoactive substances, a highly favorable effect on the psychic and somatic functions of senile patients was achieved.
Note: another study found Merinex also treated patients with mood and anxiety disorders.1,2
A Chilean study evaluated 100 patients with cerebrovascular diseases (e.g., a previous stroke, cerebral embolism, or hardening of the arteries of the brain), many of whom were senile, who received DMSO orally and through intramuscular injections over the course of 50 days. It was noted that their coronary heart disease and high blood pressure had a good improvement in 74.35% of DMSO recipients, a fair response in 21.77%, and no response for 3.88%.1,2 The neurologists overseeing these patients remarked that:
“Recovery from the general symptoms was positive; there were favorable changes which were reflected in a feeling of well being, the recovery of agility, changes of mood from depressed to gay [cheerful], improvement of sleeping, and clearer speech. As regards the ‘focal’ results, accelerated recovery from hemiplegia and hemiparesia was registered. A speedier recovery of speech in cases of defined or indicated aphasia took place.”
Additionally, in 127 elderly patients (aged 75–85) with cerebrovascular disorders, bitemporal electrophoresis with Neuromidin in a DMSO solution resulted in clinical improvement by the 5th procedure in 86%, including normalized sleep, reduced dizziness, improved memory and hearing, and decreased cognitive disorders (versus delayed improvement in controls receiving standard therapy alone). In another study of 250 patients with cerebral vascular pathology, DMSO used as a solvent for vinpocetine in cervical-collar zone electrophoresis stabilized brain blood flow parameters and improved blood supply to the large cerebral arteries. Likewise, a Russian geriatrics physiotherapy monograph recommended DMSO in transcranial electrophoresis protocols for discirculatory encephalopathy (chronic cerebral ischemia), including 50% DMSO solutions with aspirin. In a clinical study of 154 patients undergoing open revascularization for acute lower limb ischemia, intravenous DMSO (10 mg/kg) administered before reperfusion as part of a comprehensive prevention protocol resulted in reduced reperfusion injury markers, fewer renal dysfunction cases, less cardiopulmonary worsening, and milder cognitive issues compared to standard care.
Note: at a Russian military sanatorium treating victims of terrorist attacks and other emergencies, 25% DMSO was among the most frequently used agents for neurological injuries (primarily spinal pain and cerebrovascular disease), most commonly as part of a 21-day electrophoresis rehabilitation course (that was noted to effectively resolve pain).
Reader Reports
A variety of readers have reported cognitive improvements from DMSO:
I had a series of small strokes in August of 2023. I’m using 1 tsp of 99% DMSO twice a day and the results are remarkable! The improvement in mental acuity is amazing and the whole body pain relief is the best I’ve ever experienced.
I had ‘brain fog’ for 6–8 weeks after a total hip job which I am pretty sure was from the anesthetic, started drinking 2 tsp DMSO in 8oz water ~6–7 weeks after the op and feel this totally cured it in about 1 week and now my brain is very much back to normal.
I used to have the kind of mind that once skated through an Ivy League degree. Thanks to your writings on DMSO and Walsh’s nutritional therapy the last couple weeks I have begun to feel the fire that used to burn inside of my mind, the brilliant processing ability I used to take for granted. My brain actually works!!
The only thing that’s come close to helping with my Moderna vax injury. Gastroparesis, extreme brain fog, edema, SFN, MCAS, POTS, tinnitus, insomnia, mood disturbances...all improving for the first time in 3½ years!
Several additional readers reported that topical DMSO applied to the neck improved brain fog following COVID vaccination,1,2 while others found oral DMSO cleared brain fog from zolpidem, statins, chronic fatigue syndrome, a TBI, fluoroquinolone toxicity, or long-haul COVID symptoms.1,2
Memory
In addition to brain health (vibrant circulation and minimal inflammation or cells being trapped in the cell danger response) being intertwined with sleep, both are also intertwined with mental health, cognition and memory. As such, I have received a few memory improvement stories1,2,3,4,5,6 like these:
MWD, your stack is transformative. We now make your zeta potential formula, and have noticed observable results in a short time, it is a daily routine now. We also incorporated DMSO (topically) and marvel at its ability to repair and heal (and yes we have noticed the Lucid dreaming!) - these two items are the basis of our ‘first aid’ kit that travels wherever we go. Thank you for passing on this knowledge. Inadvertently we started ‘studying’ post Covid, reading and listening to a vast amount of information - and more times than not falling asleep in the process. This article explains why we can now seem to retain so much information in our 60s than we ever did in our younger days.
I am a Family Physician who was diagnosed with MCI by Mass General Neurology. I responded by cleaning up my diet, exercising daily, and taking a bunch of supplements, including daily oral DMSO. My memory is now better than it was decades ago.
So many benefits & people have noticed…Memory is insanely good now.
DMSO alone is generally thought to have a minimal impact here (e.g., one paper stated “In all experiments, dimethyl sulfoxide (DMSO) was used as vehicle, since it has no significant effect on passive avoidance learning and memory”) so a variety of studies exist where DMSO facilitated another agent improving memory.
Studies which have combined a therapeutic agent with DMSO to enhance learning and memory are as follows:
In three different rat studies, pemoline (Pm) or magnesium pemoline (MgPm) was dissolved in DMSO to influence memory and learning. In one, where rats were evaluated on how many trials it took them to learn a complex maze, compared to a saline baseline, the following reductions were observed: DMSO alone (11.1%), Pm (25.3%), low dose MgPm (32.5%), high dose MgPm (44.6%)—whereas amphetamine worsened learning by 14.5%.1,2 In a second study, MgPm (both low and high dose) dissolved in DMSO completely prevented retrograde amnesia induced by electroconvulsive shock (ECS). Lastly, a third study found these agents did not improve reversal learning (the ability to cognitively update an ingrained habit when the reward rule changes).
Note: DMSO was also shown to increase transport of pemoline into the brain.
Additional agents enhancing learning or memory in otherwise healthy or aged animals include pregnenolone⬖ (which improved spatial learning and upregulated choline acetyltransferase in aged rats), capsaicin⬖ (which improved acquisition and retention and mitigated a CB1/CB2 agonist's negative effects on learning), rapamycin (which improved novel object recognition and spatial working memory in middle-aged mice while protecting hippocampal neurons), testosterone (which nearly doubled hippocampal dendritic spine density via ERK/MAPK signaling and restored spatial memory in gonadectomized rats),1,2 nandrolone (which improved spatial learning and long-term potentiation in adolescent rats), and intracerebroventricular DHEA⬖ (which enhanced memory retention and prevented amnesia). Datumetine modulated hippocampal and prefrontal NMDA receptor signaling, upregulating CREB phosphorylation and BDNF expression, while sumac extract⬖ enhanced passive avoidance learning and memory retention. In C. elegans, a mitochondrial uncoupler preserved mechanosensory neurons, short-term memory, and extended lifespan. Aniracetam and 1-BCP reversed pyrilamine-induced working memory deficits and restored hippocampal theta power.
These therapeutic agents (in DMSO) showed strong protective effects against memory impairment across a variety of injury models. Following strokes, 4-methylumbelliferone reduced infarct volume and improved learning and memory by downregulating HAS1/HAS2, modulating inflammatory cytokines, and reducing oxidative stress. In aged rats, pioglitazone improved learning and memory by reducing oxidative stress and increasing antioxidant enzymes in the hippocampus and cortex. In Alzheimer’s disease model rats, oral quercetin⬖ significantly improved spatial learning and memory while lessening hippocampal oxidative stress and neuronal damage. 5-HT2A receptor agonists protected long-term memories from being disrupted by new memories formed shortly afterward. In ovariectomized rats modeling hormonal loss, an ERβ agonist reversed spatial learning and reference memory deficits along with impairments in dendritic spine density and synaptic structural integrity, while estrogen supplementation partially restored spatial learning and significantly increased CD147 expression (a neuroprotective protein involved in synaptic function and amyloid-β clearance). Additional agents protecting against memory impairment include Artemisia absinthium⬖ (which reversed scopolamine-induced deficits and counteracted oxidative stress), a cannabinoid agonist (which prevented scopolamine-induced spatial memory impairment), a selective CB1 antagonist (which improved consolidation of aversive associative memory), and 7,8-dihydroxyflavone⬖ (which restored spatial learning in rats subjected to maternal separation stress).
Note: in studies detailed in other parts of this article, memory and learning improvements were also demonstrated by agents combined with DMSO across a wide range of injury models — including chronic cerebral hypoperfusion (fructose 1,6-diphosphate⬖), chronic restraint and psychological stress (necrostatin-1, flupirtine, and retigabine), sleep deprivation (quercetin⬖ and almorexant), sepsis-associated encephalopathy (an LRRK2 inhibitor), manganese neurotoxicity (sodium para-aminosalicylate), anesthesia exposure during pregnancy (suberoylanilide hydroxamic acid), hyperthyroidism (AMPA and NMDA receptor agonists), and NMDA-receptor blockade modeling glutamatergic dysfunction (Ptychopetalum olacoides⬖) — collectively reinforcing that the cognitive benefits of DMSO-delivered therapies extend across virtually every category of neurological insult that impairs memory.
Lastly, in a study showing that impairing dural (meningeal) lymphatic drainage disrupts spatial working memory and interhemispheric coherence, six weeks of daily DMSO injections had no adverse effects on memory.
Movement Disorders
Movement disorders encompass conditions characterized by abnormal voluntary or involuntary movements — including tremors, ataxia (loss of coordination), dystonia, and muscle spasticity. Many arise from dysfunction in the cerebellum, basal ganglia, or their neural connections, areas particularly sensitive to the circulatory and inflammatory disruptions DMSO addresses (e.g., consider the previously mentioned Lurcher mice study where DMSO counteracted cerebellar damage). For example:
In a copper-deficient sheep model, a single subcutaneous injection of DMSO combined with copper sulfate during mid-pregnancy completely prevented enzootic ataxia (swayback) in lambs (0% incidence vs. 60% in untreated controls), performing comparably to multiple oral copper doses and suggesting DMSO enhanced systemic copper delivery sufficiently to protect fetal neurological development. In veterinary case reports, IV DMSO contributed to the recovery of a mare with ataxia and cranial nerve deficits from temporohyoid osteoarthropathy (near-normal neurological status by discharge on day 5), while in a foal with cerebellar abiotrophy, DMSO as part of a multi-agent regimen produced temporary neurological improvement before the animal relapsed due to the progressive nature of the underlying genetic condition. In another horse, DMSO was used to manage vestibular disease and blindness that developed following a jugular catheter complication during anesthesia recovery.
In rats, a neurotensin antagonist (in DMSO) attenuated neuroleptic-induced vacuous chewing movements (a model of tardive dyskinesia), supporting a role for neurotensin in its pathogenesis, while abscisic acid⬖ (in DMSO) improved harmaline-induced tremor and motor performance.
Reader reports suggest DMSO can benefit several movement disorders. One reader with vaccine-induced Stiff Person Syndrome reported that topical 100% DMSO was the only treatment that relieved constant muscle spasms — spanning the back, calves, feet, and chest — over 22 months of uncontrolled pain, noting “I have it with me at all times.” A 74-year-old reader with essential tremors, multiple spinal and thoracic fractures, and advanced tendonitis credited “DMSO, and DMSO alone” with keeping them out of a wheelchair. Other readers reported improvements in essential tremors, in tremors from vaccine injury and a neck tremor that responded to topical DMSO.
Several readers also reported that topical DMSO dramatically improved restless leg syndrome, in some cases allowing them to discontinue long-term medications they had taken for decades.
Additionally, multiple dog owners reported that topical DMSO applied to the temples or behind the ears rapidly resolved vestibular episodes — stroke-like events characterized by disorientation, wobbly legs, and vomiting — with one describing recovery within an hour and continued preventive use whenever symptoms reappear, while another credited DMSO (along with grounding) with giving their elderly dog an extra year and a half of life.1,2
Seizures and Epilepsy
Epilepsy is a neurological disorder characterized by recurrent seizures (sudden bursts of abnormal electrical activity in the brain that can cause involuntary movements), altered consciousness, and in severe cases, prolonged seizures that cause permanent brain damage or death. DMSO’s ability to cross the blood-brain barrier, counteract oxidative stress, modulate ion channels, and suppress excitotoxic glutamate signaling suggest it could help epilepsy — though as detailed below, its effects are notably dose-dependent.
Anti-Seizure Properties
Several studies have directly examined DMSO’s effects on seizure activity, revealing a consistent biphasic pattern: therapeutic doses suppress seizures while high doses can provoke them. In rats genetically prone to epilepsy, low-dose DMSO (1.65 mg/kg) significantly decreased the number and total duration of spike-wave discharges, while high doses (825–1651 mg/kg) significantly increased them — with all effects being fully reversible (supported by the fact higher concentrations of DMSO begin blocking GABA-induced chloride currents). Likewise when 50% DMSO was injected into the brain’s ventricles (diluting to approximately 6% in the CSF), no seizure activity was generated over the next hour, whereas 75% and 100% did create a significant spike in mice hippocampal evoked potentials.
In a different temporal lobe epilepsy model, high-dose DMSO (1651 mg/kg) produced significant anticonvulsant effects — prolonging seizure onset latency by 32%, shortening seizure duration by 34%, and reducing afterdischarge duration by 45% — effects the authors attributed to suppression of glutamatergic NMDA/AMPA-mediated calcium influx. In mice with chronic temporal lobe epilepsy, only 100% DMSO (1651 mg/kg) reduced seizure number and cumulative duration (by 19–41% depending on sex), while lower concentrations had no effect — and notably, DMSO did not alter acute seizure threshold in non-epileptic mice, suggesting its anticonvulsant action is specific to the chronically epileptic brain. Another study confirmed this pattern, finding 10% DMSO significantly reduced PTZ-induced epileptiform activity while 100% DMSO increased it. DMSO also raised seizure thresholds by approximately 9% against two different seizure-inducing agents and prolonged the latency to convulsions from hyperbaric oxygen exposure (thereby reducing seizures).
In specific contexts, DMSO has shown targeted protective effects: it partially prevented convulsions induced by 5-aminolevulinic acid (reducing their number and duration), likely through hydroxyl radical scavenging, and pretreatment with DMSO has been shown to prevent iron-induced epileptiform discharges, suggesting a potential role in preventing post-traumatic epilepsy.
Note: the biphasic dose response is an important consideration when using DMSO either therapeutically or as a research vehicle in epilepsy studies. Several studies have specifically documented proconvulsant effects: DMSO prolonged cortical epileptic afterdischarges in immature rats (most pronounced in the youngest animals), lowered tonic seizure thresholds in a PTZ model, and in one study, even 0.1% DMSO accelerated PTZ kindling and potentiated hippocampal neuronal damage. These findings underscore the importance of dose selection and appropriate vehicle controls in epilepsy research.
Additionally, DMSO’s ability to open the blood-brain barrier creates a dual-edged property: when excitatory amino acids (L-aspartate or L-glutamate) were administered peripherally in DMSO, they induced seizures they would not otherwise cause — which while a safety consideration, has proven useful for studying NMDA receptor involvement in seizure circuits and for demonstrating that the kindled amygdala plays a critical role in generalized seizure expression.1,2,3,4
Seizure Reduction
The most direct clinical evidence for DMSO reducing seizures comes from patients with Niemann-Pick disease type C (detailed earlier). In NPC patients, oral DMSO decreased seizure frequency and improved EEG findings. Most notably, in an 8-year-old girl with frequent seizures and severe psychomotor deterioration, two years of oral DMSO decreased seizure frequency enough to allow tapering of an anticonvulsant, produced marked EEG improvement (with normalization of theta waves and spindle morphology), and halted the progression of cortical atrophy.
In veterinary medicine, IV DMSO has been used as part of multimodal seizure management. In a horse that developed seizures following surgery, IV 10% DMSO (1 g/kg twice daily for four days) — administered for its free radical scavenging and thromboxane-inhibiting properties to maintain cerebral blood supply — contributed to seizure cessation by day 3 and a satisfactory long-term outcome. In a 2-day-old foal with perinatal asphyxia presenting with seizures, cerebral edema, and acute renal failure, IV DMSO contributed to marked resolution of cerebral edema and complete neurological recovery by day 12. In a 3-day-old foal with convulsions and ataxia secondary to electrolyte derangements from bladder rupture, IV DMSO was included in supportive care that resolved the neurological signs within days.
Lastly, as mentioned above, a dog which had seizures develop from hydrocephalus had those seizures improve from IV DMSO.
Combination Studies
A large number of agents combined with DMSO have demonstrated anticonvulsant or neuroprotective effects in seizure models.
Among those directly reducing seizure severity, glibenclamide produced the greatest effects: following status epilepticus, it significantly reduced brain edema, blood-brain barrier damage, and neuronal loss while more than doubling 28-day survival (47.8% vs. 22.2%). Resveratrol⬖ prolonged seizure latency, shortened seizure duration, reduced the brain injury marker S100B in cerebrospinal fluid and serum, protected hippocampal CA1 and CA3 neurons, and improved spatial learning and memory in PTZ-kindled rats.1,2 Resveratrol⬖ also protected against hyperbaric oxygen-induced convulsions. Quercetin⬖ (10–20 mg/kg) significantly prolonged seizure onset, reduced severity, and shortened generalized seizure duration in PTZ-induced seizures, though higher doses (40 mg/kg) lost efficacy and in a picrotoxin model paradoxically shortened seizure onset. The DMSO combination of palmitoylethanolamide (PEA) with the neurosteroid ganaxolone markedly amplified seizure suppression and eliminated mortality, whereas PEA alone showed no efficacy in that model (though it did exhibit anticonvulsant activity in kindled amygdaloid seizures).1,2 Montelukast synergistically enhanced phenobarbital’s anticonvulsant effect (when both were dissolved in DMSO), lowering the effective dose while reducing phenobarbital-induced sedation. Ruxolitinib shortened seizure duration, lowered seizure frequency over 4 weeks, and improved memory. A p38MAPK inhibitor prolonged seizure latency by 45% and halved seizure frequency. A 5-HT6 receptor antagonist reduced spontaneous recurrent seizure frequency, increased protective potassium channel (KCNQ2/3) expression, and in combination with ERK1/2 or Fyn inhibitors reversed the aberrant mossy fiber sprouting that perpetuates chronic epilepsy.1,2 Triptolide⬖ inhibited epilepsy in mice by improving hippocampal GABAergic inhibition and reducing IL-1β levels.
Carvedilol suppressed spontaneous seizure activity in hippocampal slices from Alzheimer’s model mice — a finding with implications for the seizure susceptibility frequently seen in that disease.
In a high-throughput screening of 343 essential oils using a PTZ-induced zebrafish epilepsy model, 52 demonstrated antiepileptic activity, with 15 (including patchouli⬖ and cinnamon oils⬖) showing potency far exceeding that of phenytoin. Maslinic acid,⬖ identified through computational gene expression profiling, showed significant antiepileptic activity in the same model and selectively inhibited voltage-gated sodium channel subtypes Nav1.2 and Nav1.7.
Many agents combined with DMSO also protected against the neuronal damage that follows status epilepticus. Mitochondrial division inhibitor 1 (Mdivi-1) repeatedly reduced neuronal apoptosis and oxidative stress across multiple studies, in some cases also increasing seizure latency, decreasing seizure frequency, and improving post-seizure cognition.1,2,3,4,5 Recombinant human erythropoietin protected hippocampal neurons via PI3K/Akt anti-apoptotic signaling across multiple seizure models.1,2 Ganoderic acid⬖ reduced hippocampal damage through multiple pathways (caspase-3 reduction, CaSR/JNK/P38 modulation, and improved dendritic spine density and spatial memory).1,2,3 Salidroside⬖ dose-dependently increased seizure latency, enhanced antioxidant defenses, and reduced endoplasmic reticulum stress-mediated apoptosis. Gastrodin⬖ reduced seizure severity, protected hippocampal neurons, enhanced GABAA receptor expression, and regulated post-status epilepticus autophagy.1,2 Rolipram rescued cognitive deficits, hippocampal long-term potentiation, and CREB phosphorylation in immature rats following status epilepticus. Curcumin⬖ decreased aberrant mossy fiber sprouting through miR-134/LIMK1 modulation. ERK1/2 and p38 MAPK inhibitors reduced inflammatory markers, chemokine expression, and microglial activation in the hippocampus of immature rats with kainic acid-induced epilepsy.
Additional agents showing neuroprotective effects in status epilepticus models include glycyrrhizic acid⬖ (enhanced mitochondrial autophagy and antioxidant defenses in juvenile epilepsy rats), glycyrrhizin⬖ (reduced HMGB1/p38MAPK in the hippocampus), an Omi/HtrA2 inhibitor (ucf-101; increased anti-apoptotic XIAP and HAX-1), levetiracetam (dose-dependently reduced oxidative stress and caspase-3 in epileptic offspring mice), a caspase-12 inhibitor, olomoucine (a CDK inhibitor that reduced neuroinflammation and apoptosis), 2-methoxyestradiol (via HIF-1α suppression), a CaMKII inhibitor, honokiol,⬖ a hydrogen sulfide donor (via PI3K/Akt), an IRAK1/4 inhibitor, an exosome inhibitor (which prolonged seizure latency by reducing BBB permeability changes), an eEF2K inhibitor that restored AMPKα1 expression and improved mitochondrial function, cognition, and social behavior in chronic epileptic mice, and a PARP-1 inhibitor (DPQ) that reduced neuronal apoptosis and activated the PI3K/Akt-SIRT1 survival pathway.
Interestingly, GPER-1 (the G protein-coupled estrogen receptor) activation with G1 improved spatial learning, memory, and reduced mossy fiber sprouting in chronically epileptic rats, yet the same receptor’s activation with G-1 or estradiol increased acute seizure susceptibility and nitric oxide levels in cortex and hippocampus during PTZ kindling — suggesting estrogen-mediated effects on epilepsy differ between acute seizure generation and chronic post-epileptic recovery.
Natural compounds demonstrating anticonvulsant effects in zebrafish or rodent models include vitexin⬖ (comparable to diazepam), sesamin⬖ (via PI3K/Akt), onopordia⬖ (via the nitric oxide/nNOS pathway), linalool⬖ and trans-nerolidol⬖ (with nerolidol⬖ showing superior efficacy), the combination of ursolic acid⬖ with caprylic acid,⬖ Lippia sidoides extract⬖ (comparable to diazepam), olive pit polyphenols⬖ (which protected against kainic acid-induced neurotoxicity), a tryptamine derivative of securinine⬖ (with antioxidant, chelating, and anticonvulsant properties), ε-viniferin⬖ (which inhibited NLRC4 inflammasome activation in astrocytes), thymoquinone,⬖ and progabide (which specifically suppressed the tonic phase of seizures). Lamotrigine combined with anakinra partially normalized psychoneurological disturbances (anxiety, locomotor activity, social behavior) in chronically epileptic rats, suggesting their combination addresses both seizure control and behavioral comorbidities.
Rapamycin combined with pitolisant markedly improved anxiety (2.7-fold on maze testing) and depression (37.5% improvement in swim testing) in PTZ-kindled epileptic rats, highlighting that DMSO-delivered agents can address the psychiatric comorbidities that frequently accompany epilepsy.
In stimulant toxicity models, MK-801 dissolved in DMSO abolished EEG afterdischarges and seizures induced by cocaine, amphetamine, and methamphetamine — though notably, eliminating seizures did not prevent death from methamphetamine or cocaine, demonstrating that seizure-independent toxicity pathways drive stimulant lethality.
Note: CBD⬖ in DMSO blocked voltage-gated sodium channels and demonstrated anticonvulsant effects in a PTZ model, whereas cannabigerol produced comparable sodium channel blockade but had no anticonvulsant effect — indicating that sodium channel inhibition alone does not confer seizure protection. 3-carene⬖ modulated resting-state brain activity but did not reduce epileptiform activity. Retigabine, despite being an established antiepileptic drug, paradoxically increased spike-and-wave discharge number and duration in aged WAG/Rij rats, revealing age-dependent pro-epileptic effects from neuronal potassium channel activation. Two plant-derived extracts (Searsia dentata⬖ and Searsia pyroides⬖) inhibited NMDA receptor currents and reduced intracellular calcium influx, supporting their traditional use for epilepsy.
DMSO and Antiepileptic Drug Interactions
Several findings are relevant to using DMSO alongside conventional antiepileptic drugs. DMSO has been safely used as a vehicle for diazepam in seizure treatment studies. The antiepileptic drugs carbamazepine and lamotrigine dissolved in DMSO showed no concerning interactions on human reproductive tissue, suggesting compatibility during pregnancy. A pharmacological study demonstrated favorable molecular interactions between levetiracetam and DMSO-water mixtures, supporting DMSO’s potential utility in antiepileptic drug formulation. Mood-stabilizing antiepileptics (zonisamide, carbamazepine, valproate) dissolved in DMSO enhanced monoamine and acetylcholine systems in the striatum and hippocampus at therapeutic doses while reducing them at supratherapeutic doses — a biphasic pattern that may partly explain both their therapeutic effects and certain side effects. DMSO combined with ethosuximide may synergistically increase brain epinephrine levels, as DMSO alone can stimulate the central nervous system and increase catecholamines, potentially augmenting the neurotransmitter changes the antiepileptic produces. Menthol⬖ dissolved in DMSO demonstrated dose-dependent anticonvulsant effects, with 400 mg/kg significantly reducing seizure activity below control levels — while simultaneously counteracting the proconvulsant influence of high-concentration DMSO, suggesting menthol⬖ may be a useful adjuvant when DMSO is used as a seizure medication vehicle.
Note: multiple studies confirmed DMSO at vehicle concentrations had no independent effect on seizure parameters, supporting its safety as a solvent at appropriate doses.1,2,3 A study employing DMSO-based electron spin resonance spectroscopy demonstrated that seizures induce rapid accumulation of ascorbate (an antioxidant) in the hippocampus in parallel with seizure progression — likely as a response to excessive free radical production during excitotoxicity — providing mechanistic support for why antioxidants like DMSO can be neuroprotective in seizure contexts.
Encephalitis
In addition to mitigating experimental autoimmune encephalomyelitis DMSO also mitigates brain inflammation triggered by a variety of infectious sources.
Viral Encephalitis
Intranasal DMSO mixed with nuclease mixture was used in human patients to treat meningitis and meningoencephalitis caused by acute viral respiratory infections.1,2
Multiple papers have noted that DMSO was used in horses to treat myeloencephalopathy from herpes, but no data was provided other than longer courses of DMSO being used in more severe outbreaks.1,2 In the only case report I’ve found, 500 mg/kg of IV DMSO (given every 24 hours to a hospitalized horse) contributed to a stabilization of neurologic deficits, clinical improvement and then full recovery.
Multiple papers have reported DMSO being used to treat suspected West Nile virus encephalomyelitis,1,2 one of which reported mild to moderate cases recovered fully after an anti-inflammatory protocol that included DMSO.
Intraperitoneal arctigenin⬖ (a plant lignan dissolved in DMSO), administered one day post intravenous infection, provided complete protection to mice from an otherwise fatal Japanese encephalitis virus infection (along with reducing brain inflammation, oxidative stress, cell death, and associated behavioral impairments).
In mice with a 100% fatal encephalomyocarditis (EMCV) viral infection, intraperitoneal DMSO combined with a DHEA derivative reduced mortality to 72.73%, greatly reduced (otherwise severe) neurological symptoms, prevented inflammatory brain lesions, and decreased brain viral load. In another study DMSO combined with a furin inhibitor or a different DHEA derivative showed strong antiviral activity against Zika and Japanese encephalitis virus in human and mosquito cells.
A p38-MAPK inhibitor dissolved in DMSO improved survival rate, reduced paralysis scores, lowered viral load in skeletal muscle, and decreased inflammatory cytokines in suckling mice with enterovirus 71 infection.
In dogs with neurological canine distemper, DMSO alone or combined with ribavirin and prednisone helped maintain higher hemoglobin levels and reduced the severity of anemia compared to regimens without DMSO. In another similar study, DMSO synergistically enhanced the treatment efficacy of ribavirin.
Parasitic Encephalitis
Equine protozoal myeloencephalitis (EPM) is a common and severe disease in horses that is difficult to treat because the medications for it have poor oral absorption. A 2009 study found that dissolving ponazuril in DMSO rather than water roughly tripled its bioavailability and enhanced its ability to reach the brain, potentially revolutionizing management of EPM.1,2 In another study, DMSO was also found to greatly enhance the absorption of toltrazuril (resulting in oral toltrazuril having 56% of the bioavailability of IV toltrazuril). In the only clinical case report I could locate, severe EPM neurological dysfunction initially improved following DMSO+ponazuril and a few other therapies, but the elderly horse eventually succumbed to the illness. Lastly, a 2014 veterinary paper noted that IV DMSO and ponazuril was a viable EPM treatment.
Megazol formulated as a gel in DMSO and combined with melarsoprol successfully cured experimental CNS-trypanosomiasis in mice.
Arctigenin⬖ improved depression-like behaviors in Toxoplasma gondii-infected mice, reduced neuroinflammation, and restored neurotransmitter balance by suppressing the TLR4/NF-κB pathway and IDO overexpression. Sertraline also inhibited Toxoplasma gondii brain proliferation, reduced neuroinflammation (TNF-α, TRAF2, NF-κB p65 nuclear translocation), and protected neurons in both in vitro and in vivo models of acute T. gondii infection.1,2
A rare complication for dogs from babesia infections is cerebellar ataxia, and in one case where this was accompanied by blindness and quadriparesis, a dose of IV DMSO and then other treatments preceded a rapid recovery.
Two species of Acanthamoeba (which can cause eye infections, and occasionally encephalitis), were found to be roughly twice as sensitive (and in one case four times as senstive) to tea tree oil⬖ when it was mixed in DMSO. The growth of another amoeba (used to model the deadly brain infecting N. fowleri) was inhibited by Torin-1 dissolved in DMSO.
Note: a review paper citing numerous 1990s veterinary textbooks stated that DMSO has been indicated in cases of viral and bacterial encephalitis, Herpes virus I, and myelitis due to protozoa [parasites].
Bacterial Meningitis
One Russian paper reported that in human patients DMSO enhanced the effectiveness of meningitis treatment protocols.1,2
In a foal with suspected bacterial meningitis, intravenous DMSO was included as part of intensive supportive therapy for its anti-inflammatory, analgesic, neuroprotective, and free radical-scavenging properties. The foal showed clinical improvement (recovery of suckling reflex and milk intake) by day 7–8, at which point DMSO was discontinued.
Sepsis-Associated Encephalopathy
When infections reach the circulation, they can frequently set off sepsis, a life-threatening inflammatory cascade. As a toxin many bacteria carry (LPS) can trigger sepsis, this condition is often studied in animals by injecting LPS.
As one of the organs harmed by sepsis is the brain (e.g., by far the most common form of encephalopathy in critically ill patients is sepsis encephalopathy) numerous studies have evaluated how agents protect the nervous system from sepsis, such as a sheep one where intravenous DMSO significantly reduced clinical signs of sepsis and lowered acute phase protein levels (haptoglobin, serum amyloid A, ceruloplasmin, and fibrinogen).
A variety of agents in combination with DMSO have also been shown to reduce LPS triggered brain injury:
•Dexmedetomidine pretreatment in rats reduced systemic and brain inflammation in sepsis-related encephalopathy by lowering inflammatory cytokines and suppressing NF-κB signaling in the hippocampus.
•Ibrutinib reduced neuroinflammation and brain damage in mice by lowering pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), increasing IL-10, and suppressing the inflammatory TLR4/AKT/PI3K/STAT3 pathway.
•Dasatinib reduced neuroinflammation and brain damage in mice by lowering multiple pro-inflammatory mediators and modulating key signaling pathways while increasing the anti-inflammatory cytokine IL-10.1,2
•Astaxanthin⬖ reduced systemic inflammation, brain injury markers, oxidative stress, and brain edema while improving antioxidant activity and histopathological damage in rats.
•Dexamethasone reduced brain and systemic inflammation and attenuated sickness behavior (hypophagia) in mice.
•Incensole acetate⬖ improved learning and memory, reduced neuroinflammation and oxidative stress, and increased antioxidant defenses and BDNF in rats exposed to LPS.
•Pioglitazone reversed rat hippocampal neurotoxicity by reducing inflammatory and apoptotic signaling (p-JNK, p-c-Jun, Caspase-3) and restoring protective pathways (p-Akt and p-p70S6K).
•Ziprasidone (in DMSO) reduced rat microglial activation and oxidative stress.
•Rapamycin dissolved in DMSO increased food intake and reduced inflammatory and hypothalamus metabolic signaling in chickens.
•An LRRK2 inhibitor reduced microglial inflammation and cell death pathways in vitro, and improved neuronal damage, microglial polarization, and cognitive performance in rats.
•Glibenclamide partially improved brain tissue pathology and myelin basic protein expression in neonatal rats with LPS-induced intrauterine infection-induced brain injury (which has clinical relevance as the study also found premature infants born to mothers with subclinical intrauterine infection had worse neurodevelopmental outcomes).
Additionally, DMSO prevented LPS-induced disruption of gap junction communication in rat astrocytes by scavenging hydroxyl radicals from peroxynitrite. Several other compounds also demonstrated protective effects in LPS-stimulated glial and neuronal models:
Icariin⬖ reduced astrocyte activation and inflammatory cytokine production (TNF-α and IL-1β) in primary astrocytes.
Genistein⬖ reduced LPS-induced TNF-α and IL-1β in BV2 microglial cells via an estrogen receptor-dependent mechanism.
Incensole acetate⬖ reduced neuroinflammation and oxidative stress, and increased antioxidant defenses and BDNF.
Chlorogenic acid⬖ lowered multiple pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-12), decreased iNOS, NLRP3, and NF-κB signaling, while increasing IL-10.
Ibrutinib suppressed nitric oxide, TNF-α, cell senescence, TLR4/NF-κB signaling, and reactive oxygen species.
Icaritin⬖ protected neurons, and in another study, astrocytes from LPS-activated glial cell-induced damage via an IGF-1R-dependent mechanism and reducing inflammatory cytokines.1,2
Asiatic acid⬖ protected primary hippocampal neurons from LPS-induced toxicity by reducing TNF-α and IL-1β, decreasing oxidative stress, and activating the Nrf2/HO-1 pathway while inhibiting NF-κB. Its protective effects were partially blocked by an Nrf2 inhibitor.
Lastly, Helenalin⬖ significantly reduced LPS-induced cranial bone destruction.
Note: in emergency brain surgeries, removed skull bones are often frozen for later reinsertion. In 39 such cases, bones frozen with DMSO showed less absorption and better outcomes than those frozen without it.
Encephalopathy
In addition to pathogens that directly cause inflammatory brain damage, a variety of other triggers can also induce encephalopathy. DMSO, alone or in combination with other agents, has shown potential benefit against many of these (e.g., this Russian paper cites DMSO as a treatment method for encephalopathy).
Ebselen (dissolved in DMSO) reduced symptoms of oxidative and nitrative stress in a rat model of hepatic encephalopathy induced by thioacetamide.
In a 2015 review, DMSO was proposed as a potential treatment for naturally occurring bovine polioencephalomalacia due to it being mechanistically plausible it could help the condition, certain veterinarians using it there with success, and a general lack of strong evidence existing for treatment options for the condition. This disease often results from a thiamine⬖ deficiency, to varying degrees responds to a repeated IVs of high-dose thiamine,⬖ and has symptomatic overlaps with Wernicke’s encephalopathy, a thiamine⬖ deficiency seen in humans (particularly alcoholics), suggesting DMSO may have use in that condition. Corroborating this, in a 2021 mouse model of thiamine⬖ deficiency, DMSO (intraperitoneal) alone partially improved behavioral deficits and reduced thalamic damage (a key region affected in Wernicke’s encephalopathy). When combined with high-dose thiamine,⬖ DMSO further enhanced recovery of locomotor function, motor coordination, and thalamic cell viability beyond thiamine⬖ alone, and helped minimize long-term neurological sequelae.
Note: DMSO’s chemical interactions with thiamine⬖ (e.g., to enhance thiamine⬖ delivery) have also been repeatedly studied.1,2,3
In a group of horses who developed leukoencephalomalacia from Fusarium moniliforme mycotoxins in their food, a DMSO combination therapy aimed at reducing cerebral edema halved their death rate (66% to 33%) and allowed the surviving horses to fully recover.
Myasthenia Gravis
In order for skeletal muscles to fire, they need to receive acetylcholine from the nerve that directs them. In myasthenia gravis (MG) the body forms antibodies to the muscle’s acetylcholine receptors (AChRs), and as they are destroyed, the muscles need more and more acetylcholine to be sent by the nerves to activate. In turn, MG is managed by various immune-suppressing medications, filtering the AChR antibodies out of the blood, and acetylcholine esterase inhibitors (which boost acetylcholine levels)—suggesting DMSO’s anti-inflammatory and AChR-augmenting properties (via acetylcholine esterase inhibition) may benefit the disease.
DMSO's potential for MG was initially discovered (accidentally) in 1980, when two researchers tested a variety of agents for their ability to reduce AChR antibodies, and realized that the DMSO being used as a vehicle for the various agents they were testing was independently reducing those antibodies. They then found giving rats daily intraperitoneal injections of 1 mL DMSO for two weeks resulted in a 52% decrease in AChR antibodies (but not total IgG levels) that persisted for an additional six weeks after treatment was terminated.1,2
Note: after this discovery, the researchers expressed their eagerness to test DMSO in humans with MG (the New York Times even covered it).
A follow-up rat study then found DMSO suppressed anti-AChR antibody levels by an average of 53%–76%, with the effect being similar regardless of whether DMSO was given orally, rectally, or intraperitoneally. Additionally, DMSO treatment suppressed the anti-AChR antibody response to a weak primary antigenic stimulus. Interestingly, when given during strong primary or secondary immune responses, DMSO instead enhanced antibody production 1.7–2.8-fold — indicating bidirectional immune modulation depending on timing and stimulus strength (or DMSO’s ability to potentiate allergens).
These antibody findings were complemented by direct evidence that DMSO restores neuromuscular function. In ex vivo mouse nerve-muscle preparations where tubocurarine was used to mimic MG (reducing the strength of nerve-evoked muscle contractions), DMSO produced a rapid, dose-dependent, and sustained restoration of twitch force — with 0.1% restoring 20–30% of force and 0.75% achieving complete restoration that was sustained for over 150 minutes. Electrophysiology confirmed DMSO increased the amplitude of nerve signals at the muscle by ~25–30%, consistent with its acetylcholinesterase-inhibiting properties.
Additional studies in frog and mammalian nerve-muscle preparations confirmed that DMSO at concentrations ≤1% enhanced neuromuscular transmission through acetylcholinesterase inhibition, while concentrations above 1% began to have depressant effects in mammals, and that THC (but not CBD) counteracted this restoration of neuromuscular function.1,2,3,4 DMSO has also been shown to reverse neuromuscular blockade caused by organophosphates (which poison the same acetylcholinesterase system that is therapeutically targeted in MG).1,2,3,4,5,6,
Note: this research inspired a 1982 study to determine if DMSO suppressed thyroid autoantibodies (which were experimentally induced in rats). It did, and also was found to increase the ratio of IgM to IgG plaque forming cells (which suggested a true immunoregulatory effect). In turn, some patients report that DMSO benefits autoimmune thyroiditis.
A variety of agents combined with DMSO have also shown benefit in experimental autoimmune MG models — including resveratrol⬖ (which reduced anti-AChR antibodies and protected AChR density at the neuromuscular junction), total glucosides of peony⬖ and artemisinin⬖ (both reduced clinical scores and anti-AChR antibodies while modulating regulatory T cells), atorvastatin-derived exosomes,1,2,3 and a caspase-1 inhibitor (which suppressed disease progression via IL-1β/IL-17 pathways).
Separately, sepsis-induced disruption of acetylcholine receptor clustering on muscle cells was reversed by GSK3β inhibition, restoring the receptor aggregation needed for normal neuromuscular transmission.1,2 In a rat sepsis model, rapamycin similarly improved nerve conduction velocity, muscle action potential amplitude, and survival by restoring acetylcholine receptor homeostasis.
Sadly, while physicians have highlighted DMSO’s potential for human patients, no human studies have ever been performed for DMSO with MG. However, one reader with generalized MG reported that after starting oral and topical DMSO in 2022, her muscle fatigue, cognitive function, and vision dramatically improved, and she has not had a myasthenic crisis since. She noted that higher doses at night would wake her after an hour feeling alert and functional — which she attributed to DMSO boosting her acetylcholine levels — and described the effect as “better than the pyridostigmine I used to take 6x/day.” Another reader with generalized MG alongside multiple other autoimmune conditions reported no myasthenic crises since starting DMSO in 2022, along with dramatically reduced muscle fatigue, greatly improved cognition and near-normal vision — after having been on 30 prescription medications, she is now nearly off all of them.1,2
Hydrocephalus
Cerebrospinal fluid (CSF) is primarily produced and circulates within chambers in the brain called ventricles. When too much CSF is produced or its drainage or flow from the ventricles is obstructed (or absorption is impaired), the ventricles enlarge and exert pressure on surrounding brain tissue. This can lead to cognitive impairment (including dementia-like symptoms), impaired coordination or gait disturbance, and urinary incontinence. Since DMSO facilitates fluid circulation and drainage, it could potentially help hydrocephalus.
Unfortunately, research in this area is quite limited, with the primarily referenced studies being work from a team that found injuring the ventricles (by puncturing them and then injecting DMSO) caused hydrocephalus in a dose dependent manner, with 2% DMSO causing a small increase (8-16% developed it, on average increasing ventricle volume by 50%), and 10% or 100% causing it much more frequently. This led the authors to postulate the effect they observed could account for why hydrocephalus is occasionally seen from other therapies which inject high concentrations of DMSO into the cerebral circulation.1,2,3
As these concentrations are much higher than what can be reached with standard DMSO infusions (let alone any other route of administration) and interventricular injections of a variety of substances besides DMSO reliably cause hydrocephalus, it is unlikely these results are relevant to DMSO users, especially if high-dose IV DMSO infusions are avoided.
Outside of this study, two case reports suggested DMSO can benefit hydrocephalus:
•In a Shih Tzu dog which had severe acute neurological symptoms (e.g., seizures and opisthotonus) due to non-communicating hydrocephalus, IV 10% DMSO (1g/kg over 50 minutes), greatly improved symptoms (e.g., seizures were largely suppressed and consciousness was restored), which the authors attributed to DMSO reducing intracranial pressure and protecting neurological tissue. Ultimately, the dog died after 50 days, but its prognosis greatly exceeded what had been expected.1,2
•A diffuse cerebral encephalopathy associated with hydrocephalus and cholesterinic [cholesterol] granulomas in a horse partially improved from a DMSO-containing regimen. In a horse with a large intracranial epidermoid cyst causing ataxia, head-pressing, and circling, IV DMSO with glucocorticoids and penicillin produced gradual clinical improvement (though the animal relapsed over six months due to the progressive nature of the lesion).
While in contrast, in a study of experimentally induced hydrocephalus where DMSO was used to deliver an aquaporin 4 inhibitor (which worsened hydrocephalus) intraperitoneal DMSO (at an unspecified dose) did not provide a therapeutic benefit.
Lastly, one reader shared that his brother had a progressive neurological disorder (syringomyelia, astrocytoma in the cervical spine causing paralysis, complications from its complex removal, and then hydrocephalus) which his (renowned) neurologists eventually said would be fatal in a few years (likely from encephalitis), so he sought out Stanley Jacob who agreed to treat him experimentally with DMSO. This proved to be a miracle for him and his spinal cord, with his neurologists saying “they’d never seen anyone in a similar situation stabilize and that was ‘making medical history.’” He lived 30 more joyful years, 20 of which he was Stanley Jacob’s patient, but unfortunately, IV DMSO gave him the greatest benefit and he was no longer able to obtain it once he stopped being Jacob’s patient.
Note: another reader, who had a 9-year-old daughter with cerebral palsy (and a shunt for hydrocephalus) shared that topical DMSO helped the cerebral palsy symptoms.
Psychiatric Conditions
Let’s now look at the rest of the data which has accumulated for using DMSO in psychiatry and note how often it mirrors the findings of the Russian researchers.
Human Studies
The most detailed study on DMSO’s use for psychiatric patients was conducted at a Peruvian psychiatric hospital, where 42 patients (25 schizophrenics, 4 manic depressive psychotics, 4 alcoholic psychotics, 4 compulsive-obsessive neurotics and 5 patients with severe anxiety) were taken off all their medications then given 2-5 intramuscular DMSO injections each day (with more given to the most psychotic patients) and compared to 16 controls receiving standard care.1,2
Of the schizophrenics, all 14 of the acute cases experienced a rapid and dramatic improvement (particularly in their agitation—especially for the catatonic-paranoid patients), with all being discharged within 45 days (three having a complete recovery 15 days after admission) and not having a recurrence. To quote one of them:
“I have been out of my mind. I don’t know what happened to me. I wonder what my children are going to say.”
Of the 11 chronic schizophrenics, 4 periodically needed hospitalization and had a complete remission following DMSO (allowing them to be discharged much faster than normal), and in those who later relapsed, there was again a positive response to DMSO. The remaining 7 were more severe cases (e.g., they had been hospitalized for over 6 years and failed years of therapies) and experienced an improvement from DMSO, but it was not enough to leave the hospital.
Note: results like this (I’ve seen similar ones with other therapies as well) lead me to believe that the existing understanding of schizophrenia is far from complete. To further support that contention, this author also shared a case of a severely delusional paranoid schizophrenic responding to DMSO.
The 4 manic-depressive psychotics (who were in the manic phase, averaging 15 days of psychomotor agitation) rapidly calmed down and lost their mania after DMSO (with their recovery being much faster than what they’d previously experienced from conventional therapy).
The 4 alcoholic psychotics (2 with hallucinations and 2 with delirium tremens) had previously been hospitalized for these issues. They rapidly responded to DMSO, with restlessness improving in the first few days while the hallucinations took longer.
The remaining patients (obsessive-compulsive neurosis and severe anxiety) also had a good response to DMSO (e.g., they were calmer, ideas did not upset them as before, they were able to act in a more spontaneous way, and they were able to overcome their obsessive compulsions).
Note: the authors of the 1967 paper noted that DMSO had antipsychotic and antianxiety properties and that its action differed from tranquilizers in that little sedation or central depression was produced. A later 1992 paper proposed that the antipsychotic effects observed in this trial resulted from DMSO suppressing excessive interleukin-2 (IL-2) production by T-lymphocytes, a mechanism also attributed to certain antipsychotics in the same model.
Sadly (like many things in medicine) these 1967 results elicited minimal interest and no attempts were made to replicate them (although a 1982 Russian review noted DMSO could be used for psychiatric disorders and a 2006 Russian literature review noted intramuscular injections of 50% DMSO had a sedating effect on psychotic patients).
To the best of my knowledge, only five other human psychiatric studies have been done with DMSO:
•In 17 patients (ages 28-55 years) with chronic depression (for at least 5-20 years) that did not respond to antidepressant therapy whose most recent depressive episode lasted 8 months to 2 years, DMSO was able to treat their depression. Specifically, the existing basic antidepressant therapy (amitriptyline, pirasidol, anafranil), was combined with 1ml 50% oral DMSO and taken three times a day, resulting in 14 (82.3%) of the patients having a resolution of their depression which persisted for the 1-4 years of follow-up each patient received.
Note: studies evaluating DMSO in combination with SSRI antidepressants are quite rare. In one of the only ones I’ve found, they jointly decreased rat appetite for sugar, while in the other (a heart study) the detected effects occurred at much higher concentrations than the blood of an SSRI user will reach.
•A study of 210 women with exacerbated chronic generalized periodontitis found that 10% DMSO mixed with 0.2% oxymetacil and delivered via ultrasound effectively relieved their periodontitis and lowered their anxiety.
•In two Chilean studies, a DMSO combination with amino acids⬖ was used to treat depressive neuroses (e.g., mood and anxiety disorder) while in another, when used to treat cognitive impairment and dementia, those patients also experienced a significant improvement of their mood (going from depressed to happy).1,2
•In women with post partum depression (PPD), blood tests consistently showed significantly lower vitamin D, serotonin, and norepinephrine. When vitamin D⬖ in DMSO was given to cultured hippocampal neurons, beneficial increases were observed in both their proliferation and secretion of serotonin, and norepinephrine (likely via the PI3K/Akt pathway).
Note: In lymphoblastoids from women with PPD, brexanolone (an approved PPD therapy) in DMSO modulated the expression of 98 genes potentially linked to PPD (which was believed to account for its rapid effect on PPD).
However, a variety of animals ones have been and will be summarized (all of which, unless otherwise specified were conducted with rats or mice).
PTSD
•In one study designed to model PTSD (via traumatic stress in adolescence), the methyltransferase inhibitor Unc0642 (dissolved in DMSO) alleviated (otherwise permanent) anxiety, depression, social and cognitive dysfunction and normalized PTSD impaired brain development.
•In a modified single prolonged stress (SPS) model of PTSD, a moderate dose of Cannabis sativa leaf extract⬖ (dissolved in DMSO) significantly enhanced the efficacy of exposure therapy in extinguishing the conditioned fear memory. The benefit was CB1 receptor-dependent and persisted in a drug-free test 14 days later.
Stress
In addition to the stress which causes PTSD, many rodent studies have modeled a variety of other stressful situations (which frequently cause anxiety or depression). These comprise the majority of DMSO psychiatric studies, and collectively, these results help support the theory of psychiatric illness proposed by the previously mentioned Russian Researchers.
•In chronic “social defeat” stress induced by repeated exposure to an aggressive male which attacks another rodent into submission, DMSO combined with Dihydromyricetin⬖ significantly improved learning and memory, decreased immobility time, and reduced anxiety. In another study, Epothilone D, dissolved in DMSO, prevented brain changes in the prefrontal cortex and hippocampus caused by this type of stress. Naringenin⬖ attenuated social defeat-induced neurobehavioral deficits, reduced oxidative stress (brain MDA), restored glutathione, and lowered TNF-α and IL-1β.
Numerous studies have shown promise for mitigating the effects of chronic emotional-painful stress (typically three weeks of EPS).
•In one,1,2 DMSO prior to the stress, increased superoxide dismutase (SOD) activity in brain homogenates and serum, in another, DMSO also increased SOD activity and decreased ceruloplasmin-transferrin activity1—suggesting DMSO’s ability to neutralize chronic stress relates to DMSO reducing oxidative stress.
•In another, DMSO prior to chronic EPS completely prevented the development of gastric ulcers, blocked the stress-induced behavioral changes in the open field test (excessive locomotion, rearing, and reduced center exploration indicative of anxiety), and normalized the cardiovascular and autonomic responses, including the hypertension and altered heart and respiration rates that occurred both during stress and during subsequent immobilization. Additionally, like the previous studies, it markedly increased SOD activity in the brain.1,2
Note: since DMSO interfered with the formation of malonic dialdehyde (a marker of lipid peroxidation) the investigators were unable to also measure if it reduced its formation within the brain.
The controversial forced swimming test (FST) evaluates depression through the shift from active coping behaviors to passive immobility in mice facing an inescapable stressor. Either alone or in combination with the tail suspension test, studies found DMSO in combination with the following substances elicited anti-depressant activity: flavonoids from Alpinia oxyphylla,⬖ cinnamon extract,⬖ asiatic acid⬖ (comparable to midazolam), trans-resveratrol,⬖ chrysin⬖1,2 (which was further enhanced by prozac and similar in efficacy to diazepam), ethanolic extract of Ptychopetalum olacoides,⬖ essential oil of Nepeta rtanjensis⬖ (a monoamine oxidase inhibitor), Riparin III⬖ (from an Amazonian plant), methanolic extracts of Cuminum nigrum (L)⬖ and Centratherum anthelminticum⬖ (which also reduced anxiety), ganoderic acid A,⬖ methanolic extract of Withania qaraitica⬖ (similar in efficacy to citalopram or desipramine), oridonin,⬖ alarin, ODQ and 7-NI, Prozac, and a cannabinoid CB1 receptor agonist.
Chronic restraint stress is a common rodent test for modeling human depression, anxiety. For CRS rats, in combination with DMSO:
•A caspase-1 inhibitor reduced the immobility, social avoidance and anhedonia which followed CRS and social defeat stress.
•Xiao Yao San⬖ and a c-Jun (JNK) inhibitor repeatedly improved anxiety-like behaviors. Additionally, the Chinese herbal formula reduced phosphorylated JNK, JNK, and P-c-Jun protein and mRNA expression in hippocampus comparably to the JNK inhibitor in CRS rats with liver depression and spleen deficiency syndrome.1,2
•Intraperitoneal quercetin⬖ significantly mitigated anxiety- and depression-like behaviors in CRS rats who also had restricted cerebral blood flow (via bilateral carotid artery stenosis) along with alleviating hippocampal demyelination, restoring myelin sheath thickness, reducing brain inflammation (via reduced IL-1β/TNF-α, increased IL-10/IL-4, promoted microglial M2 polarization) and enhanced microglial phagocytosis (elimination) of harmful myelin fragments.
•A corticotropin-releasing hormone type 1 receptor antagonist prevented stress-induced decreases in body weight, sucrose preference, and locomotion along with hypothalamus BDNF and GAP-43 upregulation (as unlike in the hippocampus, hypothalamic increases are maladaptive).
•Safranal⬖ protected against restraint stress-induced oxidative brain damage and alleviated stress-induced behavioral changes.
•Saikosaponin A⬖ and paeoniflorin⬖ relieved corticosterone induced inhibition of autophagic flux in PC12 cells by reducing LC3II/I, P62, and mTOR expression, suggesting a role in chronic stress.
Depression
The Chronic Unpredictable Mild Stress (CUS) test mirrors the development of depression in humans is one of the top tests for evaluating it in rodents. Natural compounds, targeted pathway inhibitors, and clinically used pharmaceutical drugs (administered in combination with DMSO) showed the following beneficial effects in CUS-exposed rats:
•A ptychopetalum olacoides⬖ extract prevented anxiety and hyperglycemia.
•Intraperitoneal curcumin⬖ ameliorated depressive-like behavior and upregulated (neurotrophic) BDNF, PSD-95, synaptophysin, p-Limk1, and p-cofilin expression in the prefrontal cortex. In another study, it also reversed CUS behaviors and upregulated those three neurotrophic proteins in the lateral amygdala.
•Oridonin⬖ (from Rabdosia rubescens) reduced social anxiety and depression, improved sucrose preference, open field activity, and neuronal morphology and cell numbers in prefrontal cortex and hippocampus and suppressed the inflammatory p38 MAPK/NF-κB/NLRP3 pathway.
The JNK blocker SP600125 partially modulated the hyperactive HPA axis by significantly lowering pituitary ACTH levels and upregulating glucocorticoid receptor (GR) expression in the hippocampal CA3 region compared to the solvent control, reduced the stress-induced increase in caspase-12 (suppressing hippocampal neuronal apoptosis), reduced C-jun (a pathway often overactivated in chronic stress) and enhanced electroacupuncture’s reduction of depression.1,2,3,4
•Necrostatin-1 (which blocks inflammatory cell death) also improved depressive-like behaviors.
•Intracerebroventricular alarin reduced depression, decreased latency to feed, and restored p-ERK/ERK and p-AKT/AKT (neurotrophic/neuroplasticity) activity in prefrontal cortex.
•Agomelatine reduced TRPV1-mediated calcium influx, oxidative stress, and apoptosis in hippocampal neurons of chronically stressed rats, improving depression-related behaviors.
•Pioglitazone improved depressive-like behaviors, suppressed pro-inflammatory cytokine increases (TNF-α, IL-1β, IL-6), reduced M1/M2 microglial ratio, decreased NF-κB expression, and ameliorated peroxisome proliferator-activated receptor gamma (PPARγ) expression loss in the prefrontal cortex and hippocampus
•Quetiapine ameliorated depressive-like behavior in chronic unpredictable stress rats, increased hippocampal BDNF and phospho-ERK1/2 expression, and promoted neurogenesis (and was synergistically enhanced by combination with transcranial magnetic stimulation).
Note: DMSO alone decreased elevated caspase-12 protein expression in hippocampus (which can cause neuronal death).
Separating infant rats from their mothers produces long-term depressive-like behaviors (e.g., anhedonia, HPA-axis dysregulation, and reduced hippocampal neurogenesis) that mirror those seen in human adults with depression and chronic stress. In separated rats, melatonin⬖ dissolved in DMSO significantly increased BrdU-positive cells and lowered glucocorticoid receptor expression in the dentate gyrus, directly counteracting the depressive process.
Note: The term “depression” is used for both a psychiatric (psychological/mood) state and a CNS-induced reduction in activity and alertness (neurologic depression or depressed mentation). These are related as inflammatory or infectious diseases can affect the nervous system and cause it partially shut down, triggering symptoms that overlap with both — such as reduced alertness/mentation and mood changes (often called “sickness behavior”). In certain cases, such as a foal with brain damage from losing blood supply during childbirth, DMSO’s therapeutic activity on the nervous system also addressed the neurologic depression (improved alertness and mentation). In others, DMSO has been observed to help resolve psychiatric depression that followed an illness, likely through anti-inflammatory and neuroprotective effects on shared nervous-system pathways.
Anxiety
In zebrafish, the willingness of zebrafish to leave dark areas to enter light areas is used to assess anxiety, and in this model, DMSO alone reduced anxiety (as did DMSO in combination with the chalcone C2OHPDA).
In mice and rats, the way they navigate specific mazes (e.g., elevated plus-maze test) or cross fields are used to evaluate anxiety. In one study, DMSO alone (10%, 0.5 µL) microinjected into the DPAG (a midbrain structure) greatly reduced anxiety and increased exploratory behavior.1,2 In combination with DMSO the following agents were found to reduce anxiety: eucalyptol⬖ (primary component of eucalyptus oil), cinnamomum⬖ (a hydroalcoholic extract), marjoram⬖ (extracted with DMSO), luteolin⬖ (following acute experimental colitis), asiatic acid,⬖ Casearin X,⬖ valepotriates,⬖ 5-MeO-DMT , CCK-8, CCK₂ agonists, cannabinoid and vanilloid (TRPV1) agonists.
•A mGluR2/3 agonist dissolved in DMSO reduced anxiety in chicks stressed by brief social separation.
Note: withania somnifera leaf extract⬖ (in DMSO) protected against benzo[a]pyrene-induced neurotoxicity in zebrafish by restoring normal anxiety, improving brain antioxidant status, and reducing neuronal damage in the optic tectum.
Biological Stressors
In addition to putting animals into psychologically stressful situations, a variety of stressors that directly injure the body have also been observed to trigger anxiety and depression and to respond to therapeutic combinations containing DMSO.
Two AMPA-type glutamate receptor potentiators reduced pain hypersensitivity and depression-like behavior in rats with neuropathic and inflammatory pain.
In neuropathic pain caused by a spared nerve injury (SNI), a DNA methyltransferase inhibitor (in DMSO) significantly improved depressive symptoms and increased neurotrophic brain BDNF levels. In another SNI study, a chemogenetic DREADD inhibitor (in DMSO) alleviated both pain and depressive symptoms and in a third, rapamycin alleviated anxiety. depression and pain from an L5 SNI.
In rats with pentylenetetrazol triggered epilepsy (which consistently produces anxiety and depression), rapamycin and pitolisant (dissolved in DMSO) markedly improved anxiety (2.7 fold improvement on a maze test) and depression (37.5% improvement in swim test).
In mice with LPS induced depression induced, resveratrol⬖ in DMSO reversed their depression along with reversing neural inflammation and triggering neurogenesis (particularly within the hippocampus).
In mice with toxoplasmosis triggered depression, arctigenin⬖ in DMSO significantly reduced depressive behaviors and brain inflammation by inhibiting key pro-inflammatory pathways (TLR4/NF-κB and TNFR1/NF-κB).
In ovariectomized (OVX) mice modeling menopause, resveratrol⬖ in DMSO significantly reduced ovariectomy (ovary removal) induced anxiety and depression and neural inflammation (particularly within the hippocampus).
In mice with anxiety induced by high-fat diet, Japanese ginseng⬖ in DMSO significantly reduced anxiety-like behaviors, increased brain BDNF and synaptophysin levels, and reversed the brain’s FGF21 resistance (a key driver of the anxiety).
In mice, Naringenin⬖ improved hypoxia-triggered depression and anxiety, along with reducing brain oxidative stress and inflammation, increasing BDNF expression, and protecting amygdala neurons.
In mice exposed to X-rays after consuming saccharin (as this conditions them to avoid the sweetener), topical DMSO prior to the X-ray largely prevented this condition aversion (there was an 8% rather than 66% drop in saccharin consumption). A later study had similar results, suggesting DMSO counteracts the stress that would otherwise create the aversion.1,2
Note: lastly some of the DMSO combinations described in the previous sections were used to treat conditions directly triggered by a pharmaceutical (e.g., depression from corticosterone or anxiety from salicylates).
Psychosis
•DMSO was proposed to exert antipsychotic effects by modulating chronic macrophage activation and downstream cytokine dysregulation, particularly interleukin-2 signaling, thereby reducing schizophrenia symptoms across disease phases through immune regulation.
•MK-801, a drug with the same mechanism as psychosis-inducing phencyclidine (PCP) and ketamine, is used to induce schizophrenia in rodents. A study found that atypical antipsychotics and the Src kinase inhibitor PP1, dissolved in DMSO, significantly attenuated MK-801-induced cortical (brain) injury in rats, with the protective potency of the antipsychotics correlating with their clinical effectiveness in treating psychosis.
•In a MK-801 rat study, three different drugs that activated the α7nAChR receptor (when combined with DMSO), significantly improved schizophrenic behaviors by increasing following behavior and total interaction time, decreasing avoidance behavior time along with creating therapeutic changes within the prefrontal cortex and hippocampus (increased α7 nAChR protein expression, increased cAMP levels, decreased PDE4A and PDE4D protein expression).
•Diosmin⬖ attenuated hyperactivity, behavioral deficits, oxidative stress, and neuroinflammation in a mouse model of LPS plus ketamine-induced schizophrenia-like symptoms.
•In another MK-801 schizophrenia study, the flavanoid Fisetin⬖ (given intraperitoneally in DMSO) significantly decreased rat escape latency, increased space exploration time and platform crossings and (beneficially) phosphorylated CaMKII, ERK1/2, and CREB.
In addition to these combinations treating schizophrenia, tinospora cordifolia⬖ (an Ayurvedic herb) dissolved in DMSO demonstrated antipsychotic activaty in mice given (psychosis-inducing) amphetamines (along with curcumin⬖ in DMSO counteracting methamphetamine-induced neurotoxicity and spatial memory impairment in rats).
Note: a study (which found female rats were more prone to developing methamphetamine addictions than male rats) found modafinil dissolved in DMSO, attenuated METH-seeking behavior in both sexes. Another study found parthenolide (dissolved in 1% DMSO) partially blocked cocaine’s actions in the brain (but it is unlikely this could translate to a cocaine addiction therapy).
Sedation and relaxation are sometimes reported from DMSO use (e.g., sedation has been repeatedly observed in humans and animals at higher doses, intraperitoneal DMSO was observed to cause a decrease in spontaneous motor activity and in one large trial, 3% of DMSO users reported increased tiredness). This is likely due to DMSO increasing parasympathetic tone (due to it inhibiting acetylcholinesterase), and may partially account for DMSO’s psychiatric properties as excessive sympathetic activity plays a key role in anxiety, mania and psychosis.
Note: I find a significant portion of psychiatric issues result from excessive sympathetic activity or deficient parasympathetic tone (e.g., the “buzz” that state creates frequently creates anxiety), and likewise many (myself included) believe a significant number of health issues result from vagal (parasympathetic) dysfunction. Given that DMSO can both directly increase parasympathetic function and also heal or restore the function of nerves (along with potentially relaxing tight muscles compressing them), it is possible some of DMSO’s benefits in psychiatric conditions arise from its effect on vagal function.
Finally, like many other therapeutic agents, DMSO has also been combined with antipsychotics. For example, with intraperitoneal haloperidol, it facilitated haloperidol-induced 1.8-fold increases in striatal preproenkephalin mRNA and 1.6-fold increases in total endogenous opioid peptides (with no effect in other brain regions), increasing antipsychotic efficacy, and potentially reducing pain or antipsychotic extrapyramidal side effects.
Note: a variety of antipsychotics have used DMSO as a vehicle (e.g., this study did so with intraperitoneal olanzapine, amisulpride, quetiapine and aripiprazole, while this one did so with haloperidol, clozapine, RMI-81582 and risperidone).
Sleep
Sleep is one of the most important, yet least appreciated facets of our existence, as sleep healing the body and integrating our minds is an immensely complex process that can easily be derailed (e.g., poor sleep causes neurodegeneration, and neurodegeneration worsens sleep). Sadly, rather than support it, our medical system defaults to treating insomnia with sedatives (e.g., sleeping pills) which “put you to sleep” but also sedate the restorative process of sleep—which is tragic, as restoring healthy sleep is frequently one of the top three things which needs to be done to heal chronic illnesses.
Since DMSO heals the nervous system and restores impaired fluid circulation (which I believe is what ultimately underlies many cases of insomnia), I hence was hopeful DMSO could be a remarkable sleep aid to facilitate restorative sleep.
Yet, in the 6,000 reports I’ve received from readers, I have only received a few reports indicating that happened.1,2,3,4,5,6,7,8,9,10,11
The thing I have noticed most recently is a reduction of fatigue. I used to indulge in a daily nap, then go to bed and sleep another 8-9 hours at night. Now, if I try to nap I just can’t be bothered. I sleep my normal sleep at night and awaken refreshed.
Note: another reader had an almost identical experience, while another reported DMSO addressed the excessive sleep they had long required following a stroke.
I take a couple of mls before bed helps dramatically improve my sleep.
I feel amazing. My sleep is generally remarkable.
I love the really deep sleep it gives me. Feeling very tired after months of long intense work, I took a small dose one morning just because. An hour later I had no choice but to sleep. Woke up hours later totally refreshed. Amazing stuff.
I have started using it instead of Hydroxyzine, which I use at times. No side effects!
Note: a few readers have also reported an energizing effect from DMSO causing them to wake up after much shorter periods and feeling refreshed (which may be positive or negative).
However, in the reports I’ve received, two consistent patterns have jumped out.
First, while DMSO did not inherently function as a “sleeping pill” a large number of people reported that DMSO cured an ailment which was causing insomnia, and I have received dozens of reports from individuals stating DMSO allowing them to sleep profoundly improved their life (including cases where they had previously been suicidal). This, in turn, highlights the flaws of the symptom-based approach to medicine we practice, as many cases of insomnia which have clearly identifiable causes (that are never addressed) are instead simply “treated” with sleeping pills.
Most commonly, this was due to DMSO treating musculoskeletal pain which had kept them from falling asleep (or routinely woke them up at night). Shoulder pain resolutions (e.g., bursitis, impingements, or rotator cuff tears) 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17 were the most common, followed by low back pain (e.g., radiculopathy, disc herniations or tears, failed spinal surgery, vertebral metastasis),1,2,3,4,5,6,7,8,9,10,11,12,13,14 arthritis (e.g. rheumatoid arthritis and in the hands or toes),1,2,3,4,5,6,7,8,9 knee pain (e.g., arthritis, meniscus injury, or a sprain in a 2 year old),1,2,3,4,5,6,7 hip pain (e.g., bursitis),1,2,3,4 elbow pain (e.g., tennis elbow),1,2,3 neck pain (whiplash or degenerative discs)1,2 hand pain,1,2 jaw pain1,2 lower extremity tendopathy (e.g., gluteal)1,2 plus individual instances of sleep greatly improving following the resolution of sciatica, foot pain, a throbbing thumb, tight muscles, and blunt maxillofacial trauma (from a car throwing them into the pavement).
DMSO also resolved other types of pain that prevent sleep such as peripheral neuropathy (e.g., in the feet),1,2,3,4,5,6,7 headaches (e.g., migraines, concussions),1,2,3 cancer pain1,2 individual instances of eye pain, CRPS, and trigeminal neuralgia plus numerous resolutions of unspecified pains which had prevented them from sleeping.1,2,3,4,5,6,7,8
DMSO also improved other challenging neurological conditions to the point sleep also dramatically improved such as restless leg syndrome,1,2,3,4,5,6 Lyme disease,1,2 vaccine injuries (from Moderna or the shingles vaccine)1,2 along with individual successes with Down syndrome, demyelinating polyneuropathy, cramping fasciculation syndrome [similar to ALS and until DMSO had made the reader suicidal], an elderly dog with tics, and unspecified long term neurological issues that had prevented getting a good night’s sleep for years. For example, to quote one Lyme patient:
Then I woke one morning and was astounded to realize I had slept through the night, -[whereas] before DMSO I had been waking 3 or 4 times every night because of pain.
Finally, in addition to DMSO addressing pain and neuropathies which prevented sleep, it also addressed other issues which interfered with sleep. Most commonly this was through improved breathing, specifically by addressing sinusitis (e.g., “I had the best sleep last night, I didn’t want to get up this morning.)1,2,3 or lung issues (e.g., asthma, COPD or lung damage from burn pits in Afghanistan).1,2 One reader sent a particularly interesting testimonial, suggesting DMSO can sometimes improve nightly oxygen saturation:
I tried a little 70% on a Q-tip inside each nostril before bed to see if it would help nighttime congestion. Interesting that I get no itch or tingles from inside my nostrils…maybe due to the mucus? Well, anyway, I wear a sleep tracker ring and my oxygen level and oxygen drops have significantly improved and I am sleeping in longer periods without waking up so often! I am amazed and tried to find some research that would help explain these improvements. I found some research on sleep patterns in rodents but difficult to understand. I just wanted to let you know and thank you for sharing your knowledge. I will continue my experiment and so far every night it has improved sleep data.
Note: other issues readers reported DMSO sleep improvements with include Barrett’s esophagus, severe eczema, prostate enlargement, hypothyroidism, or an acute cold.
In short, given how necessary healthy sleep is for the nervous system (discussed further here) a case can hence be made that one of the primary ways DMSO “heals the nervous system” results from it treating the ailments that were preventing healthy sleep.
Secondly, I noticed that many readers independently shared that their dreams became more vivid or lucid after taking DMSO.1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16
Separately, after several weeks of taking DMSO internally now I have indeed noticed increased dreaming. Most of my life I have either not dreamed at all or at least had no memory of any dreams. But I have always slept very deeply and soundly. Usually falling asleep within seconds of laying down and having no memory of anything until morning. When I supplemented 5HTP for a while a few years back I started to dream a lot for the first time in my life.
Two factors can potentially explain this. First, by DMSO healing the nervous system and restoring circulation, this likely aids the neurological apparatus which facilitates dreaming. Second, two compounds are commonly used to facilitate lucid dreaming, 5HTP (as the above reader used) and galantamine (one of the few drugs I’ve ever used, primarily because of its phenomenal effects with lucid dreaming—provided you do not use an excessive dose). Galantamine is an acetylcholinesterase inhibitor, and since DMSO is also widely recognized to do so, this likely accounts for it increasing the vividness of dreams. However, the effects I’ve noticed from the two are very different (to the point it would not have occurred to me a similar mechanism was involved), so if this theory is valid, my best guess is DMSO should be viewed as a much weaker acetylcholinesterase inhibitor which only affects the dreams of a subset of the population.
There seemed to be a wide mix of attitudes towards the dreams (absolutely loving them, appreciating they can finally remember them, being neutral, or finding them challenging), with the majority being positive. These responses accurately represent that spectrum:
Picked this stuff up since reading your posts, been giving me some crazy ass vivid dreams, some even fully lucid haha! Haven’t really noticed anything else yet but the dreams alone have been worth it 13/10 would recommend.
when i drink it...after a couple days...i sleep super deep and have incredibly emotional dreams...which can be tough but i wake up feeling rested (but sad or contemplative).
Funny you should mention dreams and DMSO, since I started taking it internally my dreams are super vivid and sometimes very unpleasant! There are people popping up in my dreams now that I haven’t thought of in decades…Melatonin⬖ also caused vivid dreams, but nothing like what DMSO has been causing!
Overall my favorite dream story was probably this one:
I have to share this one thing…the first evening I applied DMSO to my neck I went to bed and in the night I heard a voice saying “THE TREES WILL HEAL YOU “. In my sleepy state I had thought, oh how nice, I do love our trees ;I live in a forest. However when I awoke it dawned on me that DMSO comes from trees and that message was about the DMSO!
Lastly, one reader shared that DMSO allowed them to dramatically lower the Xanax they needed to fall asleep, while another stated it made them much more sensitive to having their sleep disturbed by coffee or beer. This is congruent with DMSO’s known ability to potentiate benzodiazepines and alcohol and suggests thought should be taken when using these agents together, but simultaneously, given that only two readers noticed this, unlike the DMSO-alcohol issue, I am not sure how impactful it is. Additionally, two readers reported DMSO’s sleep promoting effects were enhanced by combining it with magnesium⬖ (which is plausible).1,2
Sleep Research
The limited literature on DMSO and sleep shows the following:
1. DMSO alone generally has a minimal impact on the sleep cycle. For example, a 2005 rat study found 5% and 10% DMSO administered intraperitoneally had no effects on sleep architecture, but 15% and 20% shifted deep sleep to light sleep. This indicates the doses individuals take are unlikely to affect sleep unless they are in the sensitive minority. Likewise, in a study where intraperitoneal corticotropin releasing factor was found to make rats REM sleep become Non-rem sleep, DMSO alone (at an unspecified dose) was found to have no effect on sleep architecture.
Note: one literature review cited a Russian report that one side effect of DMSO is sleep disturbance, but I have not seen this mentioned anywhere else.
2. A few references corroborate that DMSO’s healing qualities improve sleep by healing something else causing insomnia:
•In a study of 35 men with exacerbated knee osteoarthritis (and upper gastrointestinal bleeding), in addition to topical 50% DMSO (with hydrocortisone, lidocaine and potassium iodide) reducing their knee pain, it also improved their sleep (and ability to work). Specifically, on a -3 (worst) to 3 (best), scale, their baseline sleep score (0.4 ± 0.2) improved to 1.1 ± 0.4 with the DMSO combination, and to 2.8 ± 0.5 when the combination was used with physical therapy.
•In 40 patients (on average 53.7 years old) with cervical osteochondrosis complicated by shoulder myofascial pain syndrome, topical DMSO (in combination with other analgesics) and physiotherapy and acupuncture fully resolved the pain for 34 (85%), partially improved it for 6 (15%) and in all patients, there was a significant improvement in sleep.
•In a clinical guide from Uzbekistan on surgical infections of the hand and fingers, DMSO was recommended as a conservative treatment (in combination with novocaine and an antibiotic) and for abscess in the fingertip pad to prevent them from becoming painful enough to cause “sleepless nights” and require surgery. In a Ukranian dental manual, DMSO was recommended for healing (and preventing infections) of dry sockets, as it resolved the conditions and improved numerous associated things including disrupted sleep.
•In a study of patients with localized scleroderma, daily topical DMSO (and a few other therapies) significantly improved skin lesions, prevented relapse and improved emotional health, appetite and sleep.
3: Most of the existing research on DMSO for sleep involved it being a delivery vehicle that was combined with another sleep-promoting agent either to facilitate its action or make it more potent. These broke into four categories, two of which supported the observations in reader reports.
Potentiation of sleeping medications:
•Prior administration of DMSO increased the sleeping time created by pentobarbitone sodium by 78%. Conversely, another study found hexobarbital sleeping times were not altered when 2.5 g/kg of 25% DMSO were given subcutaneously to mice beforehand.
•A DMSO dissolved fraction of a Coriandrum sativum extract⬖ accelerated and prolonged pentobarbital induced sleep more than any other agent tested (including diazepam).
•A Marjoram DMSO extract⬖ and separately a Nardostachys jatamansi rhizome extract⬖ significantly increased ketamine-induced sleep duration.1,2
Modulation of circadian sleeping times (with potential utility for jet lag):
•Melatonin,⬖ which accelerated re-entrainment of the mouse’s circadian rhythm after a 6-hour phase shift and also made mice more susceptible to light-induced circadian shifts.1,2 In two other studies, Triazolam (a sleeping pill dissolved in DMSO) shifted and lengthened hamsters’ circadian rhythm, whereas DMSO alone only created a small non-significant shift.1,2 Finally, DMSO’s metabolite, MSM⬖ (in DMSO) mitigated neurobehavioral impairment, oxidative stress, and disruptions in clock gene expression in mice exposure to alcohol combined with circadian rhythm disruption.
Note: we suspect DMSO has a significant synergy with photobiomodulation, but still do not have enough data to claim anything definitive.
Improving sleep apnea:
Intraperitoneal treatment with a Chinese herbal composition of Gastrodia elata⬖ and Cinnamomum cassia⬖ (dissolved in DMSO) reduced snoring pressure and frequency in aged rats and increased activity of phrenic, recurrent laryngeal, and hypoglossal nerves, while prolonging inspiratory, expiratory, and pre-inspiratory times, suggesting modulation of upper airway neural activity.
In mice with obstructive sleep apnea, dronabinol, a vagal cannabinoid agonist, significantly reduced sleep apnea episodes when combined with 100% DMSO but not when combined with 25% DMSO (while at both concentrations, REM sleep was suppressed).1,2 Separately, Anandamide (an endogenous cannabinoid) increased sleep and adenosine levels in the basal forebrain.
In an intermittent hypoxia model of sleep apnea with rats that had heart attacks, Paxil prevented intermittent hypoxia from causing systemic insulin resistance or further damage to the injured heart (e.g., fibrosis and apoptosis was reduced).
Counteracting adverse effects of chronic sleep deprivation
In mice models, a variety of substances in combination with DMSO have been found to counteract the adverse effects of chronic sleep deprivation. Kaempferitrin⬖ (from monkfruit) increased antioxidant capacity, reduced oxidative stress, and reduced obesity. Astragalin⬖ significantly ameliorated liver oxidative stress. Siraitia grosvenorii flavonoids⬖ and melatonin⬖ increased antioxidant capacity and reduced oxidative stress and inflammation. Almorexant improved spatial learning and memory and reduced (neuroinflammatory) astrogliosis. A macrophage migration inhibitory factor inhibitor improved spatial learning and memory, and partially protected the hippocampal neurons.
Modafinil (a stimulant used to increase wakefulness in individuals with chronic sleep deprivation) significantly decreased total sleep time and increased sleep latency in English bulldogs with sleep-disordered breathing, effectively alleviating hypersomnolence (tiredness).
In rats, Paxil dissolved in DMSO (for an osmotic minipump) significantly prolonged REM sleep episode duration and reduced the number of REM sleep episodes across multiple 6-h periods (which constitutes a positive shift in chronic sleep deprivation).
Down Syndrome and Developmental Disabilities
When I initially reviewed the DMSO literature, the thing I had the most difficulty believing was reports it significantly improved Down Syndrome, as like most doctors, I assumed this was a genetic condition absolutely nothing could be done for. However, after seeing the benefits being reported in numerous clinical studies and the official Congressional transcripts, I realized my assumption may have been wrong and decided to risk my credibility by publishing that data.
Shortly after, I received these testimonials from the parents of a two-year-old girl with Down Syndrome. Her father wrote first:
We’ve been giving DMSO to our young daughter who has Down Syndrome along with extra vitamins [e.g. amino acids⬖] based on the treatment protocol from some studies. Almost immediately we noticed she was sleeping better through the night, she’s become more verbal—she suddenly seems like she wants to say words more intentionally now—and her appetite has improved substantially. She just seems more active, and that’s really awesome!
Her mother then followed up:
Even in the time since my husband made that comment, we have seen changes in our little girl. The biggest one is that she is now CRAWLING—she had seemed for the longest time like she wasn’t even interested, but now she’s doing it (she started on her 2nd birthday, in fact!). Her coordination and motor skills are steadily improving, and she is super close to sitting up on her own, something I was getting really worried about. It’s almost like she’s not even the same kid she was two weeks ago. DMSO has been a game-changer for us. I am so excited to keep learning and seeing what’s possible!
These parents’ experience echoes what was presented at a 1980 Congressional hearing convened to (unsuccessfully) pressure the FDA to end its embargo on DMSO, where testimony was given of a child with Down Syndrome having a similarly miraculous response—but over a much longer timeframe.
There, Melody Clark was discussed, who at 11 months was started on DMSO by Dr. Jacob. At that time, her legs were “just like a rag doll’s,” she couldn’t roll from her back onto her stomach, and her eyes couldn’t focus. Her initial evaluation at six months had predicted she would never progress mentally beyond age six.
After seven years on DMSO, Melody had progressed from what one physician described as “practically a vegetable state of existence” to being only mildly developmentally delayed—something her teachers had never seen in another child with Down Syndrome. Specifically, she functioned at a second-grade level (excelling in math, reading phonetically, spelling, and habitually using complete sentences), could jump rope, do somersaults, and play on a trampoline, and was quite popular with her classmates—having transformed from a child who played alone and stood near the teacher watching others into one who initiated group activities and took turns with peers. Her teacher wrote to Congress that in view of Melody’s achievements, she “would like to see DMSO given a fair testing and evaluation in helping the handicapped.”
Her dentist additionally testified that her palate, jaw, and tongue had moved significantly toward normalcy—something he had never seen in this patient population. Melody’s extremely high palate at birth was now within normal range.
Two other similar cases have also been reported:
•At 10 months of age, Bronwyn Nash (who had Down Syndrome) was frail and unable to gain weight, so her mother started her on DMSO. She began gaining weight and developed an increased awareness of the people and objects around her and then started reaching out to touch things. At 18 months, she was able to stand up, and then became able to get into her mother’s cupboards, started to feed herself, and held her water glass well. At the time a health journalist visited her at 28 months of age, she was an alert, cheerful little girl much enjoyed and well loved by her family and improving steadily.
•At 14, Billy King could walk and feed himself but had the mental capacity of a ten month old. He then began drinking milk with DMSO each morning, and two years later, had the mental capacity of a seven year old and began losing the characteristic Down Syndrome appearance.
He continued to improve and was eventually able to hold a job in a Portland bookstore.
Note: another account of Billy King’s story has a differing chronology (e.g., he started DMSO at 8 not 14, and also took the amino acid⬖ formula).
Research in turn exists to support these unbelievable anecdotes.
•In Oregon, 67 moderately or severely mentally disabled children (aged 4-17) with Down Syndrome were randomized to receive a high or low DMSO dose and then were compared to 23 similar children whose parents did not want them to receive an experimental drug.1,2 No side effects occurred and a dose dependent improvement was observed:
In Chile, 55 children with severe mental disability caused by Down syndrome (the oldest being 14) were given DMSO and amino acids⬖ by intramuscular injection or served as controls. The vials for injection consisted of DMSO along with gamma aminobutyric acid (GABA⬖), gamma amino beta hydroxybutyric acid (GABOB⬖), and acetyl glutamine⬖ (with lower doses given to those under 3 1⁄2 years of age). The children’s development was then evaluated with Gese[l scores and a massive improvement was seen in the DMSO group:
Note: this study (and the additional improvements that occurred) can be viewed here and here.
In Argentina, 13 mentally disabled children (5 severe cases, 4 moderate cases, and 4 mild ones), who did not have Down Syndrome, received a DMSO amino acid⬖ mixture (known as Merinex) three times a week by injection for 180 days (with periodic 15 day periods where the amino acids without DMSO were administered orally)
Note: other authors have reported young patients (and older ones) with learning difficulties, low intelligence, ADHD, anxiety disorders, epilepsy, nervousness, dyscalculia, dyslexia, exhaustion, and concentration problems all benefit from this protocol. Additionally, some have argued adding galactose⬖ to it enhances its efficacy.
In a 1969 study, 44 severely developmentally delayed children received the DMSO amino acid⬖ mix, with many experiencing a heightened capacity for learning in a relatively short time and over 70% having favorable responses such as “increase of the IQ, an evident and accelerated progress in basic achievements, an overall improvement of intellectual capacity, evident progress in reading, writing, and mathematics, better coordination of movements and improved manual skill, and a decrease of behavioral problems,” along with gaining better psychomotor control, no longer having anger for no reason, a general reduction of irritability, and a lessening of disobedience.
Another 1969 study gave the DMSO amino acid⬖ mixture for six months to 30 learning disabled children with language disorders (who did not have an accompanying neurological illness) and compared them to 20 controls, and observed it resulted in:
•Disappearance of mental lethargy.
•Evidence of sensorial reactions.
•Disappearance of automatic movements.
•Disappearance of inertia, passivity, and negativity.
•Growing interest and initiative in tasks and activities.
•Improvement of the physiognomic expression and of the spoken language.
•Lucid activity, group contact, and disappearance of unprovoked aggressiveness.
•Losing shyness and developing self-esteem.
•Successful training to carry out chores, to do shopping, to eat, and to dress without help, etc.
•Learning to read and to write and to do homework.
A 1976 Chile study gave DMSO and amino acids⬖ (GABA,⬖ GABOB,⬖ L-acetylglutamine,⬖ and arginine⬖) to 15 children (under 3.5 years old) with Down Syndrome and compared them to 13 untreated controls. After 12 months of intensive treatment, per the Gesell Developmental Quotient, motor area average rose from 56 to 72, adaptive from 50 to 66, language from 52 to 58, and social from 40 to 64 (with many individuals improving >10 points, especially in motor, adaptive, and social domains), while controls remained largely unchanged. Physical improvements included reduced macroglossia, better facial expressiveness, increased muscle tone/coordination, and postural changes in treated children. During the subsequent 6-month maintenance phase (to 18 months total), treated children’s gains were largely maintained or slightly increased (e.g., motor to 74, adaptive to 69, social to 67), with no significant regression and only minor isolated losses (e.g., in language for a few), whereas controls showed no progress or slight declines. The authors noted no significant adverse effects and concluded that early intervention appeared beneficial for neuronal stimulation and development.1,2,3
One author reported on an Argentinian study (I could not find) conducted by this physician, where 18 children with Down syndrome received DMSO and amino acids⬖ and were compared to 91 controls, and to quote the author this resulted in:
a tendency towards accelerated maturity in the children treated, with marked progress in language integration; this could be established in statistically significant degrees in the children treated
Additionally:
•This study, and this study, also found the DMSO amino acid⬖ mixture benefited developmentally disabled children.
•A 1982 Spanish literature review highlighted DMSO as a treatment for Down Syndrome.
•A 2009 Russian diabetes book promoted regularly applying a topical mixture of DMSO, hydrogen peroxide, and copper sulfate for diabetes and tissue regeneration, which cited a case of a pregnant woman whose fetus was diagnosed with Down Syndrome who did that protocol and then had a normal child.
•1980 and 1982 articles by skeptical physicians acknowledged DMSO was being explored as a treatment for Down Syndrome.
Note: in addition to Down Syndrome, the German DMSO community has had significant success using DMSO and amino acids⬖ for learning disabilities, developmental delays, mood and anxiety disorders, and neurodegenerative diseases—of which minor or severe learning disabilities are the most common application.
Autism
One of the most frequent questions I receive is if DMSO can be used to treat autism (as many factors mechanistically argue for it), but unfortunately, other than a few vague parent reports I’ve come across suggesting it worked (I could not corroborate), I do not have any direct parental data to assess this question.
However, a variety of studies on mice (modified to model autism) have found DMSO in combination with another therapeutic helped their autism-like behaviors. Specifically, with DMSO:
Epothilone D (a microtubule-stabilizing agent) improved autism-like repetitive behaviors across multiple studies1,2,3,4,5,6 through a consistent mechanism: stabilizing microtubules, increasing excitatory synapse number, and enhancing myelin basic protein expression in the cerebral cortex. These studies consistently found reduced self-grooming times and increased social ability, with individual studies additionally demonstrating improved restricted repetitive behaviors in marble burying tests, normalized myelination and increased dendritic spine density.
Curcumin⬖ repeatedly enhanced sociability, reduced repetitive behaviors, and restored hippocampal neurogenesis across multiple studies,1,2,3,4 with consistent findings of increased hippocampal neuron counts and reduced excessive astrocytes (a key facet of the disease process), along with improved learning, memory, and social communication. Individual studies further showed curcumin⬖ promoted neural progenitor cell proliferation, increased radial glial cells in the subgranular zone, and enhanced newborn neuron maturation.1
Prenatal resveratrol⬖ prevented embryonic valproic acid exposure from causing autism-like behavioral features, including deficits in nest-seeking behavior, social interaction, and over-excitation in the brain’s glutamate system.1,2
Melatonin⬖ and Wortmannin (a PI3K inhibitor) improved autism-like behaviors, promoted synapse-associated protein synthesis, and improved dendritic spine development. Separately, Wortmannin alone rescued dendritic spine density, upregulated postsynaptic density protein, and inhibited the same pathway rapamycin targets.
Additional agents improving autism-like behaviors in mouse models include a 5-HT2A receptor antagonist (which attenuated repetitive self-grooming), a S1PR1 antagonist (which improved learning and memory while preserving hippocampal neuronal density and microvascular integrity), estradiol (which restored empathy and social affection in ovariectomized mice), and baicalin⬖ (which dose-dependently improved recognition and spatial memory while increasing BDNF and healthy neuron counts in the hippocampus).
Collectively, these studies using a diverse variety of therapies that share the common thread of DMSO suggest DMSO may also independently improve autism, but due to DMSO rarely, if ever, being compared to a saline placebo in these types of studies, it is impossible to know.
Note: complete summaries of each study can be found in my reference Substack here.
Other Developmental Disorders
Cerebral Palsy: Given DMSO’s ability to heal central nervous system damage, particularly from temporary loss of blood flow (along with its ability to help muscle spasticity), it appears plausible DMSO could help with cerebral palsy. Unfortunately, this has not been studied and the only paper I’ve found on this topic noted that DMSO with verteporfin partially restored the ability of muscle satellite cells to correctly differentiate, potentially reducing their spasticity.
However, in horses, one paper reviewing veterinary textbooks reported DMSO treats neonatal brain damage (e.g., hypoxic-ischemic encephalopathy) and colic in foals, while another book reported that veterinarian Jack Metcalf had found horses developmentally disabled at birth (to the point they can’t nurse) once given IV DMSO three times daily regain the ability to nurse and that DMSO accelerates their overall development.
Finally, one of the most common and disabling features of cerebral palsy is severe disabling muscle spasticity (to the point aggressive measures like botox are routinely used). In patients with severe muscle spasticity, Russian researchers reported success with ultrasound-facilitated topical applications to the affected muscles and applicable spinal segments of DMSO mixed with sodium oxybutyrate (GHB, the potent sleep aid the FDA went to war against) and lidocaine. In the detailed patent (which I translated here), they specifically discussed a case of a patient who developed debilitating spasticity from arachnoiditis, but implied it could be used to regain mobility in other conditions like poliomyelitis or cerebral palsy. In parallel, one reader with a cognitively intact nine-year-old girl with cerebral palsy that affects three limbs shared that her daughter told her topical DMSO relaxed her tight muscles and allows her to move more easily.
Note: another Russian patent proposed DMSO as a transdermal enhancer for a botox patch to treat the spasticity associated with Parkinson’s, cerebral palsy, dystonia, and multiple sclerosis — potentially bypassing the need for the repeated injections botox currently requires. Outside of this patent, I have only seen DMSO botox formulations used for refractory idiopathic detrusor (bladder) overactivity (where they worked).
Cleft Palate: One of the original anti-seizure medications, phenytoin, was notorious for causing a pattern of birth defects, some of which (such as a flat nasal bridge and epicanthal folds) can resemble features seen in Down syndrome. One study found DMSO prevented some of the birth defects (e.g., cleft palate) phenytoin caused.
ACBD6-related neurodevelopmental syndrome: this rare disease also causes cognitive impairment and birth defects resembling those seen in Down Syndrome. While DMSO has not been tested on patients with it, the disorder is caused by defective activity of N-myristoyltransferase, an enzyme DMSO has been shown in vitro to increase the activity of.
Krabbe disease: this is a rare incurable genetic lysosomal storage disease that causes severe neurodegeneration and severe developmental delays at the start of life. A 1999 paper disclosed that Stanley Jacob had developed and implemented an oral DMSO protocol for this condition but it is unclear if it was successful.
Tourette syndrome: in disease-model rats, Vitamin D⬖ (1.0 μg/kg/day) in DMSO significantly decreased locomotor behaviors (0.43–0.69 vs 0.92–1.33 scores) and reversed the decrease in striatal dopamine content (along with causing an expected vitamin D⬖ induced increase in serum calcium).
Note: a lot of people have asked if DMSO could help muscular dystrophy. The only data I’ve found is a study where (-)-epicatechin⬖ in DMSO reduced dystrophic fiber pathology and promoted muscle regeneration in MD mice, but did not address the underlying disease mechanism — which unfortunately tells you very little.
A New Model of Neurology
Through reading this, my hope is that it has not only given you hope for treating many “incurable” neurological conditions, but also that you share my fascination in the new perspective it provides on what ultimately causes many of these diseases.
In turn, while this article was lengthy, you may have noticed many neurological conditions were omitted. This was due to the length of this article (due to all its links) crashing Substack and the article hence needing to split into multiple parts. So, in addition to previous articles I’ve written which show DMSO provides similar neurological benefits to each of the sensory organs (e.g., particularly the eyes and ears), in the upcoming parts of this series I will cover the remarkable effects DMSO has provided for:
•Spinal issues (e.g., paralysis, spinal cord injuries, disc issues, radiculopathies, and arachnoiditis).
•Peripheral nerve issues (e.g., nerve regeneration, palsies, neuropathies and neuropathic pain).
•Strokes, brain bleeds, traumatic brain injuries, and concussions.
In the final part of this article, I will share practical guidance for properly obtaining each grade of DMSO (including IV DMSO), provide specific protocols for the neurological conditions discussed here — including Parkinson’s, Alzheimer’s, strokes, chronic stress, neuropathies, cognitive impairment, brain fog, anesthesia toxicity, and neurodevelopmental disorders — along with highlighting complementary natural therapies we have found to further benefit these conditions.”